The Complete ATAC-seq Protocol: A Step-by-Step Guide from Nuclei Isolation to Data Analysis for Researchers
This comprehensive guide provides researchers and drug development professionals with a detailed, up-to-date explanation of the Assay for Transposase-Accessible Chromatin using sequencing (ATAC-seq) protocol.
The Complete ATAC-seq Protocol: A Step-by-Step Guide from Nuclei Isolation to Data Analysis for Researchers
Abstract
This comprehensive guide provides researchers and drug development professionals with a detailed, up-to-date explanation of the Assay for Transposase-Accessible Chromatin using sequencing (ATAC-seq) protocol. Covering the full workflow from foundational principles to advanced applications, the article explores the biochemical basis of the assay, offers a meticulous step-by-step methodological breakdown, addresses common troubleshooting and optimization challenges, and discusses validation strategies and comparisons with other epigenomic techniques. Designed to be a practical resource, it equips scientists with the knowledge to successfully implement ATAC-seq in their own research to map chromatin accessibility and decipher gene regulatory landscapes.
What is ATAC-seq? Understanding Chromatin Accessibility and Epigenetic Landscapes
Core Concepts of Chromatin Architecture
Chromatin architecture refers to the multi-scale organization of DNA and its associated proteins within the nucleus. This structural hierarchy is fundamental to regulating genomic functions such as transcription, replication, and repair. The dynamic interplay between compacted and accessible chromatin states dictates cellular identity and function.
Nucleosomes are the fundamental repeating unit of chromatin, consisting of approximately 147 base pairs of DNA wrapped around an octamer of core histone proteins (H2A, H2B, H3, and H4). Nucleosomes compact the genome and serve as a regulatory platform through post-translational modifications (histone PTMs) and histone variant incorporation.
Open Chromatin refers to genomic regions where nucleosomes are depleted, displaced, or structurally altered, making DNA more accessible to transcription factors (TFs), RNA polymerases, and other regulatory machinery. These regions are often associated with active regulatory elements like promoters, enhancers, and insulators.
Gene Regulation is directly controlled by chromatin architecture. The positioning and stability of nucleosomes at transcription start sites (TSSs) can block or permit the assembly of the pre-initiation complex. Conversely, accessible chromatin facilitates TF binding and transcriptional activation.
Quantitative Data on Chromatin States
The table below summarizes key quantitative features associated with different chromatin architectural states.
Table 1: Quantitative Features of Chromatin Architectural States
| Architectural Feature | Typical Genomic Size | Key Histone Modifications | Associated DNA Feature | Approximate Frequency in Human Genome |
|---|---|---|---|---|
| Nucleosome Core Particle | ~147 bp DNA wrap | H3K4me1, H3K27ac (Promoter); H3K9me3, H3K27me3 (Repressed) | - | ~30 million nucleosomes / diploid cell |
| Linker DNA | ~20-60 bp | - | - | - |
| Open Chromatin Region (e.g., ATAC-seq peak) | 100 - 1000 bp | H3K27ac, H3K4me3, H3K4me1 | Transcription Factor Binding Sites, DNase I Hypersensitive Sites (DHS) | ~100,000 - 200,000 peaks per cell type |
| Active Promoter | 500 - 2000 bp | H3K4me3 (high), H3K27ac, H3K9ac | CpG Islands, TATA Box, Initiator (Inr) | ~20,000 - 25,000 per cell |
| Active Enhancer | 500 - 5000 bp | H3K27ac, H3K4me1, H3K122ac | Mediator complex binding, Cluster of TF motifs | ~50,000 - 100,000 per cell type |
Experimental Protocols for Chromatin Accessibility Mapping
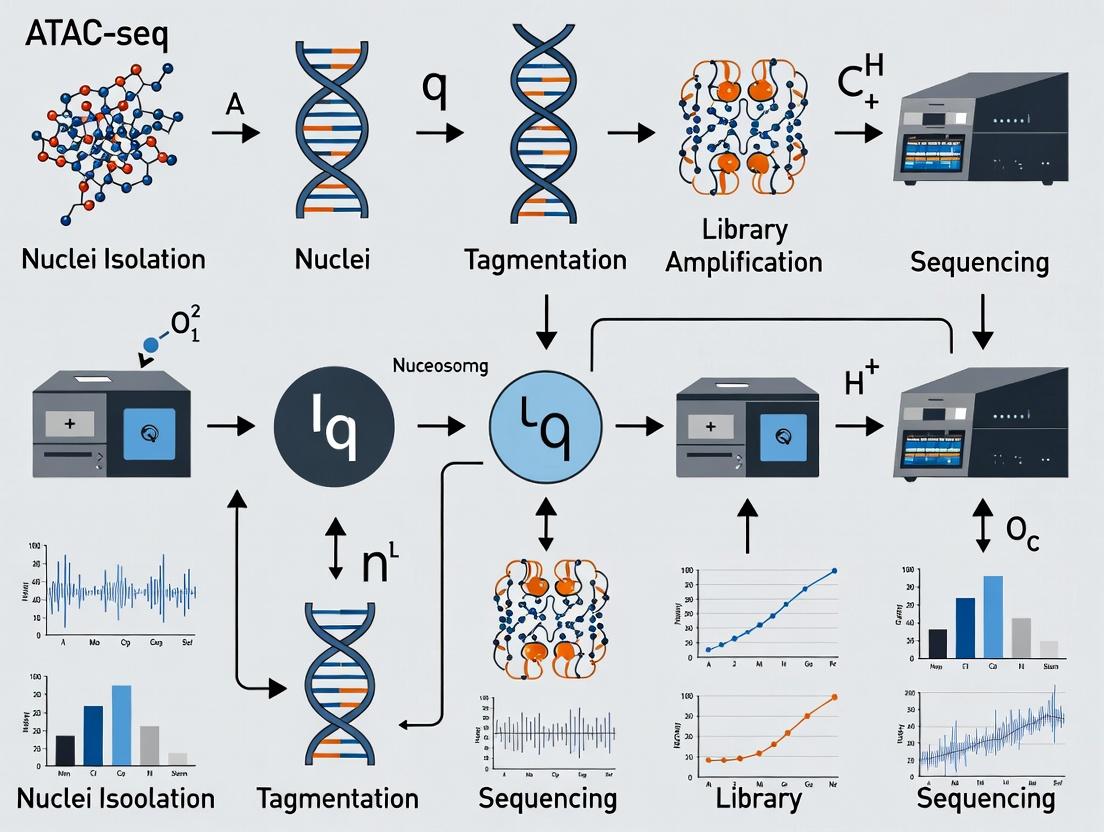
Understanding chromatin architecture requires experimental methods to probe nucleosome positioning and accessibility. The following is a detailed protocol for the Assay for Transposase-Accessible Chromatin with high-throughput sequencing (ATAC-seq), a core technique within the thesis context.
Detailed ATAC-seq Protocol
Principle: A hyperactive Tn5 transposase inserts sequencing adapters directly into open, nucleosome-free regions of the genome. The tagged DNA fragments are then purified, amplified, and sequenced.
Reagents and Equipment:
- Cell lysis buffer (10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl2, 0.1% IGEPAL CA-630)
- Transposition reaction mix (25 µL 2x TD Buffer, 2.5 µL Tn5 Transposase, 22.5 µL Nuclease-free water)
- DNA purification beads (SPRI beads)
- Thermocycler
- Bioanalyzer/TapeStation
- High-throughput sequencer (e.g., Illumina)
Step-by-Step Workflow:
- Nuclei Isolation: Harvest 50,000 - 100,000 viable cells. Pellet cells and resuspend in cold lysis buffer. Incubate on ice for 3-10 minutes to lyse the plasma membrane while keeping nuclear membranes intact. Pellet nuclei at 500 x g for 10 minutes at 4°C.
- Transposition: Resuspend the nuclear pellet in the transposition reaction mix. Incubate at 37°C for 30 minutes in a thermocycler with shaking.
- DNA Purification: Immediately clean up the transposed DNA using SPRI beads. Elute in a small volume (e.g., 20 µL) of elution buffer or water.
- PCR Amplification: Amplify the purified DNA using a limited-cycle (typically 5-12 cycles) PCR program with barcoded primers. The optimal cycle number is determined by a qPCR side-reaction to avoid over-amplification.
- Size Selection and Cleanup: Perform a double-sided SPRI bead cleanup to remove large fragments (>1000 bp, multi-nucleosomes) and very small fragments (<100 bp, primer dimer). This enriches for fragments representing nucleosome-free regions (~100-300 bp) and mono-nucleosome-protected fragments (~200-600 bp).
- Library QC and Sequencing: Assess library quality and fragment size distribution using a Bioanalyzer. Quantify the library and sequence on an appropriate platform (e.g., Illumina NextSeq, 2x 50 bp paired-end reads).
Data Interpretation: Sequencing reads are aligned to a reference genome. The distribution of fragment sizes shows a periodicity of ~200 bp, reflecting nucleosome patterning. Peaks in the insertion site track represent regions of open chromatin.
Diagrams of Workflows and Relationships
ATAC-seq Experimental Workflow
Chromatin Folding and Functional States
The Scientist's Toolkit: ATAC-seq Research Reagent Solutions
Table 2: Essential Reagents and Materials for ATAC-seq Experiments
| Item | Function/Description | Key Considerations |
|---|---|---|
| Hyperactive Tn5 Transposase | Engineered enzyme that simultaneously fragments and tags accessible DNA with sequencing adapters. | Commercial kits (e.g., Illumina Tagment DNA TDE1) ensure high activity and lot-to-lot consistency. |
| Cell Permeabilization Detergent (e.g., IGEPAL CA-630) | A non-ionic detergent used to lyse the cell membrane while keeping nuclei intact. | Concentration and incubation time are critical to prevent nuclear lysis. |
| SPRI (Solid Phase Reversible Immobilization) Beads | Magnetic beads that bind DNA fragments for purification and size selection. | The bead-to-sample ratio determines the size cutoff for selection, crucial for enriching nucleosome-free vs. mono-nucleosome fragments. |
| High-Fidelity PCR Mix with Unique Dual Index Primers | Amplifies the tagmented DNA library while adding sample-specific barcodes for multiplexing. | Limited-cycle PCR is essential to prevent skewing representation. Index primers allow pool sequencing. |
| Fluorometric DNA Quantification Kit (e.g., Qubit dsDNA HS) | Accurately measures low concentrations of double-stranded DNA library. | More accurate for library quantification than spectrophotometry (Nanodrop), which is sensitive to contaminants. |
| High-Sensitivity DNA Bioanalyzer/TapeStation Kit | Assesses the final library's fragment size distribution and quality. | Confirms the characteristic ~200 bp periodicity pattern and absence of adapter dimer. |
Assay for Transposase-Accessible Chromatin using sequencing (ATAC-seq) has revolutionized the study of chromatin architecture and gene regulation. At the heart of this protocol lies the engineered Tn5 transposase, a molecular tool that simultaneously fragments and tags genomic regions based on their physical accessibility. This whitepaper deconstructs the core biochemical principle of Tn5, framing it as the critical first step in the broader ATAC-seq workflow. Understanding this mechanism is paramount for researchers, scientists, and drug development professionals aiming to interpret epigenetic landscapes in disease and development.
Biochemical Mechanism of the Hyperactive Tn5 Transposase
The wild-type Tn5 transposon is a composite transposon from E. coli. For ATAC-seq, a hyperactive mutant (e.g., E54K, L372P) is used, which exhibits reduced sequence specificity and increased catalytic rate. The core principle is its ability to perform "cut-and-paste" transposition in vitro.
The Catalytic Core: The Tn5 transposase functions as a dimer. Each monomer binds to a specific 19-bp mosaic end (ME) sequence that is part of the engineered transposon DNA. In ATAC-seq, this transposon DNA is pre-loaded with adapter sequences, creating a "loaded transposome" complex.
Targeting Mechanism: Tn5 does not have an inherent sequence-based targeting mechanism for open chromatin. Instead, its targeting is purely physical and steric. The ~100 kDa transposome complex can only efficiently access and insert into genomic DNA that is not compacted into nucleosomes or bound by other proteins. Nucleosome-bound DNA is sterically hindered, preventing transposase integration. This physical exclusion is the fundamental principle that maps regulatory regions.
Tagging (Integration) Reaction: The loaded transposome performs a series of concerted DNA cleavage and strand transfer reactions:
- Synapsis: The dimer brings the two adapter-bearing MEs together.
- Double-Strand Cleavage: The transposase cleaves both strands of the genomic DNA target site, creating a 9-bp staggered overhang.
- Covalent Integration (Tagging): The 3’-OH ends of the cleaved genomic DNA attack the phosphodiester bonds at the 3’ ends of the transposon (adapter) DNA. This results in the covalent ligation of the adapter sequences to both ends of the generated genomic fragment.
This "tagging" simultaneously fragments the accessible DNA and appends universal priming sequences for subsequent PCR amplification and sequencing.
Diagram Title: Tn5 Transposome Cut-and-Paste Integration
The efficiency and bias of Tn5 transposition are critical parameters for ATAC-seq data quality.
Table 1: Key Quantitative Metrics of Tn5 Transposition in ATAC-seq
| Metric | Typical Value/Range | Significance & Impact on Assay |
|---|---|---|
| Catalytic Rate (k~cat~) | ~10 s⁻¹ (hyperactive mutant) | Determines required incubation time; faster kinetics reduce assay time. |
| Integration Site Bias | ~9 bp periodicity in vitro | Reflects DNA helical pitch; can create non-uniform coverage patterns. |
| Fragment Size Distribution | Peaks <100 bp (nucleosome-free), ~200 bp (mono-nucleosome), ~400 bp (di-nucleosome) | Directly maps chromatin accessibility and nucleosome positioning. |
| Genomic DNA Input | 50,000 - 100,000 nuclei (standard) | Lower input increases technical variability; higher input improves signal-to-noise. |
| Transposase to DNA Ratio | Critical optimization point | Excess Tn5 causes over-fragmentation; insufficient Tn5 yields low library complexity. |
| Reaction Time | 30 min - 1 hour at 37°C | Must balance complete tagmentation with minimal mitochondrial DNA contribution. |
| Insert Size (Stagger) | 9 bp | Defines the "duplication" on complementary strand after gap repair/PCR. |
Detailed Experimental Protocol: In Vitro Tagmentation
This protocol details the core Tn5 reaction as performed in a standard ATAC-seq workflow.
Objective: To fragment accessible genomic DNA and ligate sequencing adapters simultaneously using a pre-loaded Tn5 transposase.
Materials & Reagents: See "The Scientist's Toolkit" below.
Procedure:
- Nuclei Preparation: Isolate cells of interest. Lyse plasma membranes using a cold lysis buffer (e.g., 10 mM Tris-HCl, pH 7.4, 10 mM NaCl, 3 mM MgCl₂, 0.1% IGEPAL CA-630). Pellet nuclei at 500-1000 x g for 10 min at 4°C. Resuspend nuclei in a known volume of cold PBS.
- Quantification: Count nuclei using a hemocytometer or automated cell counter. Adjust concentration.
- Tagmentation Reaction Mix: In a nuclease-free PCR tube, combine the following components on ice:
- 25 µL 2x Tagmentation Buffer (provided with commercial kits, typically containing Mg²⁺)
- Up to 50,000 nuclei in a volume of 20 µL (diluted in PBS)
- 5 µL of loaded Tn5 transposase (commercially available, e.g., Illumina Tagment DNA TDE1 Enzyme)
- Nuclease-free water to a final volume of 50 µL.
- Incubation: Mix gently by pipetting. Immediately incubate the reaction at 37°C for 30 minutes in a thermal cycler with heated lid (105°C) to prevent evaporation.
- Reaction Arrest: Add 25 µL of Stop Buffer (40 mM EDTA, 200 mM NaCl, 1% SDS, 2 mg/mL Proteinase K). Mix thoroughly.
- Cleanup & Elution: Incubate at 40°C for 30 minutes to digest transposase and proteins. Purify the tagged DNA using a MinElute PCR Purification Kit or equivalent SPRI bead-based cleanup. Elute in 20 µL of low-EDTA TE buffer or nuclease-free water.
- Library Amplification: The eluted DNA contains adapters on both ends. Amplify with 10-12 cycles of PCR using primers compatible with the transposon-adapter sequences and containing full Illumina flow cell adapters and sample indexes. Purify the final library.
Diagram Title: Core ATAC-seq Tagmentation Workflow
The Scientist's Toolkit: Essential Reagents & Materials
Table 2: Key Research Reagent Solutions for Tn5 Tagmentation
| Item | Function & Role in the Core Principle | Example/Note |
|---|---|---|
| Engineered Hyperactive Tn5 Transposase | The core enzyme. Pre-loaded with sequencing adapters to form the active transposome complex. | Illumina Tagment DNA TDE1 / TDE1, Diagenode Hyperactive Tn5, or custom in-house expression/purification. |
| 2x Tagmentation Buffer | Provides optimal ionic strength (Mg²⁺ is an essential cofactor) and pH for transposase activity. | Typically supplied with commercial Tn5; contains MgCl₂, DMF, etc. Critical for efficiency. |
| Cell Lysis Buffer | Gently lyses the plasma membrane while keeping nuclear membrane intact, releasing nuclei for tagmentation. | Contains Tris, NaCl, MgCl₂, and a mild non-ionic detergent (e.g., IGEPAL CA-630). |
| Stop Buffer | Halts the tagmentation reaction by chelating Mg²⁺ (EDTA), denaturing proteins (SDS), and digesting Tn5 (Proteinase K). | Prevents over-fragmentation and prepares sample for DNA purification. |
| SPRI (Solid Phase Reversible Immobilization) Beads | Magnetic beads that bind DNA for purification and size selection, removing reaction components and small fragments. | Essential for cleaning up tagmented DNA and selecting optimal fragment sizes post-PCR. |
| PCR Master Mix with High-Fidelity Polymerase | Amplifies the low-quantity tagmented DNA, adding full sequencing adapters and sample-specific indexes. | Must be robust for low-input, GC-biased templates. Often incorporates NEBNext High-Fidelity 2X Master Mix. |
| Nuclease-Free Water & Buffers | Prevents enzymatic degradation of input DNA, transposomes, and final library. | A critical quality control point to avoid assay failure. |
Within the broader thesis on the ATAC-seq protocol, this whitepaper details how mapping chromatin accessibility provides a critical functional readout of the epigenome, linking regulatory DNA dynamics to fundamental biological processes and therapeutic interventions. ATAC-seq (Assay for Transposase-Accessible Chromatin using sequencing) has become a cornerstone technology for identifying open chromatin regions, enabling researchers to connect transcription factor binding, enhancer activity, and nucleosome positioning to phenotypic outcomes in development and disease, ultimately informing drug discovery pipelines.
Chromatin Accessibility as a Functional Genomic Hub
Chromatin accessibility is a dynamic regulator of gene expression. Accessible regions, devoid of condensed nucleosomes, are targets for transcription factors (TFs) and co-regulators that drive cell state-specific programs. Disruption of these patterns is a hallmark of developmental disorders and diseases like cancer.
Table 1: Quantitative Impact of Chromatin Alterations in Disease
| Disease Context | Key Chromatin Alteration | Measured Effect (Typical ATAC-seq Data) | Associated Functional Outcome |
|---|---|---|---|
| Cancer (e.g., AML) | Gain of de novo enhancers | 2,000-5,000 new accessible regions in leukemic vs. normal progenitors | Activation of oncogenic transcriptional programs (e.g., MYC, HOX genes) |
| Neurodevelopmental Disorders | Non-coding variation in accessible chromatin | ~40% of ASD-linked SNPs reside in accessible regions of developing neurons | Disruption of TF binding sites, altered gene expression in neurogenesis |
| Cardiac Hypertrophy | Reprogramming of enhancer landscape | ~12,000 regions show differential accessibility upon stress | RE-engagement of fetal cardiac gene programs |
| Inflammatory Disease | Dynamic opening at cytokine loci | Increased accessibility at IL6, TNF promoters (peak height increase >5-fold) | Amplification of inflammatory response |
Detailed Methodologies: From ATAC-seq to Insight
The following protocols outline core experiments linking chromatin dynamics to application areas.
Protocol 1: ATAC-seq in a Disease Model System
Objective: To identify differentially accessible chromatin regions between diseased and healthy control cells.
- Cell Preparation: Harvest 50,000-100,000 viable cells per condition (e.g., primary patient cells vs. healthy donor, or treated vs. untreated cell line). Use gentle centrifugation (300-500 x g, 5 min, 4°C).
- Cell Lysis & Transposition: Resuspend cell pellet in 50 µL of ATAC-seq lysis buffer (10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl2, 0.1% Igepal CA-630). Incubate on ice for 3 minutes. Immediately add 50 µL of transposition mix (25 µL 2x TD Buffer, 22.5 µL nuclease-free water, 2.5 µL Tn5 Transposase from Illumina "Tagment DNA TDE1 Enzyme") to the nuclei. Mix gently and incubate at 37°C for 30 minutes in a thermomixer with shaking (300 rpm).
- DNA Clean-up: Purify transposed DNA using a MinElute PCR Purification Kit (Qiagen). Elute in 21 µL of Elution Buffer.
- Library Amplification: Amplify the library using 2x KAPA HiFi HotStart ReadyMix and indexed primers (Nextera Index Kit). Use a qPCR side reaction to determine optimal cycle number (typically 5-12 cycles) to avoid over-amplification. Run the main reaction for the determined cycles.
- Library Purification & QC: Clean the final PCR product using SPRIselect beads (Beckman Coulter) at a 1.2x ratio. Assess library quality and size distribution (~200-1000 bp modal) using a Bioanalyzer (Agilent) or TapeStation.
- Sequencing: Sequence on an Illumina platform (typically 75-150 bp paired-end). Aim for 50-100 million reads per sample for mammalian genomes.
Protocol 2: Integration with Transcriptomics (Multi-omics)
Objective: To correlate differential chromatin accessibility with gene expression changes.
- Parallel Sample Processing: Split a single cell suspension from the same biological condition into two aliquots. One aliquot is processed for ATAC-seq (as in Protocol 1). The other is processed for RNA-seq (e.g., using a poly-A selection or ribosomal depletion protocol).
- Bioinformatic Integration:
- Process ATAC-seq data: Align reads (Bowtie2/BWA), call peaks (MACS2), identify differential peaks (DESeq2 or diffBind).
- Process RNA-seq data: Align reads (STAR/HISAT2), quantify gene expression (featureCounts), identify differentially expressed genes (DESeq2/edgeR).
- In silico linkage: Use tools like GREAT or ChIPseeker to associate differentially accessible peaks (particularly distal enhancers) with putative target genes based on genomic proximity (e.g., ±500 kb from TSS).
- Correlation analysis: Statistically test (e.g., Fisher's exact test) for enrichment of gene expression changes among genes linked to altered accessible regions.
Visualizing Signaling and Workflow Logic
Diagram 1: Core ATAC-seq Workflow to Key Applications
Diagram 2: Drug Action on Chromatin-Mediated Gene Regulation
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Chromatin Accessibility Studies
| Item | Function & Rationale |
|---|---|
| Tn5 Transposase (Tagmentase) | Engineered transposase that simultaneously fragments and tags accessible genomic DNA with sequencing adapters. Core enzyme of ATAC-seq. |
| Nextera Index Kit (Illumina) | Provides unique dual indices for multiplexing samples during library amplification, allowing cost-effective sequencing of multiple libraries in one run. |
| SPRIselect Beads (Beckman Coulter) | Solid-phase reversible immobilization (SPRI) beads for size-selective cleanup of libraries, removing primer dimers and large fragments. |
| KAPA HiFi HotStart ReadyMix | High-fidelity, hot-start PCR enzyme mix for minimal-bias amplification of tagmented DNA libraries. Critical for low-input samples. |
| Cell Permeabilization Buffer | A detergent-based buffer (containing Igepal/Digitonin) to lyse the cellular membrane while keeping nuclei intact for accurate tagmentation. |
| Nuclei Counter (e.g., Countess II) | Accurate quantification of isolated nuclei is essential for optimizing transposase input and ensuring consistent, high-quality data. |
| ATAC-seq Grade Nuclei Isolation Kits | Pre-optimized kits for specific tissues (e.g., brain, heart, frozen tumors) that provide high nuclei yield and purity, reducing background. |
| Epigenetic Modulators (Tool Compounds) | Small molecule inhibitors (e.g., JQ1 for BET proteins, Tazemetostat for EZH2) used to perturb chromatin states and validate regulatory mechanisms. |
Within a comprehensive thesis on the ATAC-seq protocol, meticulous pre-protocol planning is the cornerstone of experimental success and biological validity. The Assay for Transposase-Accessible Chromatin sequencing (ATAC-seq) requires careful upfront decisions regarding sample quality, cellular input, and replicate strategy to ensure robust, reproducible, and interpretable data. This guide details the critical planning stages preceding the wet-lab procedure.
Sample Considerations
The nature and quality of the starting material dictate the entire experimental trajectory.
Key Factors:
- Cell Type: Primary cells, cultured cell lines, or nuclei from frozen tissues each present unique challenges. Primary cells are more biologically relevant but have limited numbers and viability.
- Viability: ATAC-seq is highly sensitive to mitochondrial contamination from dead or dying cells. Viability >90% is strongly recommended.
- Sample Purity: Homogeneous cell populations are ideal. For heterogeneous samples (e.g., tumors, whole tissues), consider prior sorting (FACS) or enrichment, as mixed cell types confound chromatin accessibility signals.
Experimental Protocol for Sample Preparation:
- Cell Harvesting: Use gentle dissociation methods to minimize stress responses that alter chromatin.
- Viability Assessment: Count cells using a hemocytometer with Trypan Blue exclusion or an automated cell counter. Calculate viability:
(Viable Cell Count / Total Cell Count) * 100. - Nuclei Isolation (for certain tissues): For tough or frozen tissues, isolate nuclei using a hypotonic lysis buffer (e.g., 10 mM Tris-HCl, pH 7.4, 10 mM NaCl, 3 mM MgCl2, 0.1% IGEPAL CA-630) on ice, followed by centrifugation and resuspension in cold PBS.
- Cryopreservation (if necessary): Flash-freeze cell pellets or isolated nuclei in liquid nitrogen. Store at -80°C. Thaw on ice immediately before use.
Cell Number and Input Requirements
Optimal cell input balances data quality with practical constraints. Insufficient input leads to poor library complexity, while excess can cause over-tagmentation.
Table 1: Recommended Cell/Nuclei Input for ATAC-seq
| Sample Type | Recommended Input (Cells/Nuclei) | Key Rationale & Notes |
|---|---|---|
| Mammalian Cell Lines | 50,000 - 100,000 | Standard range for robust signal. High viability is critical. |
| Primary Cells (e.g., T-cells) | 50,000 - 200,000 | May require higher input due to larger nucleus-to-cytoplasm ratio. |
| Sorted/Purified Populations | 10,000 - 50,000 | Feasible with optimized, low-input protocols. |
| Frozen Tissue Nuclei | 50,000 - 100,000 | Assess nuclei integrity post-isolation. |
| Low-Input/Single-Cell Protocols | 500 - 10,000 | Requires specialized reagents and bioinformatics. |
Detailed Methodology for Cell Number Titration Experiment:
- Prepare a single-cell suspension with >95% viability.
- Aliquot into five tubes containing 10,000, 25,000, 50,000, 100,000, and 200,000 cells.
- Process each aliquot through an identical, scaled-down version of the ATAC-seq protocol (lysis, tagmentation, purification, amplification).
- Assess outcomes via:
- Bioanalyzer/TapeStation: Library fragment distribution.
- qPCR: Amplification curves to assess library complexity.
- Sequencing: Final data metrics (e.g., FRiP score, library complexity).
Replicate Design
Proper replication is non-negotiable for distinguishing technical noise from biological variation and for statistical power.
Table 2: ATAC-seq Replicate Design Strategy
| Replicate Type | Minimum Recommended Number | Definition & Purpose |
|---|---|---|
| Biological Replicates | 3 (ideally 4-5 for complex studies) | Genetically distinct samples from different biological units (e.g., different mice, donors, cultures). Essential for generalizability and statistical significance. |
| Technical Replicates | 2 (for assessing protocol variance) | Aliquots from the same biological sample processed independently through the protocol. Distinguishes protocol-induced noise. |
Experimental Protocol for Replicate Processing:
- Independent Harvest: For biological replicates, harvest cells from independently grown cultures, animals, or patients on different days.
- Parallel Processing: Process all replicates in parallel using identical reagent lots, equipment, and personnel to minimize batch effects.
- Randomization: Randomize the order of sample processing on the thermocycler and sequencer to avoid positional bias.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for ATAC-seq Pre-Protocol Planning
| Item | Function | Example/Notes |
|---|---|---|
| Viability Stain | Distinguishes live from dead cells for accurate counting and quality control. | Trypan Blue, DAPI (for nuclei), Propidium Iodide (flow cytometry). |
| Cell Strainer | Removes cell clumps to ensure a true single-cell suspension. | 40 µm nylon mesh strainers. |
| Nuclei Isolation Buffer | Gently lyses plasma membrane while leaving nuclei intact for difficult samples. | Contains a non-ionic detergent (e.g., IGEPAL CA-630). |
| Cell Counting Device | Accurately quantifies cell concentration and viability. | Automated Cell Counter (e.g., Countess II) or hemocytometer. |
| Cryopreservation Medium | Preserves cells/nuclei for long-term storage at -80°C. | Contains FBS and DMSO (for cells) or glycerol/sucrose (for nuclei). |
| DNA Binding Beads | Size-selects tagmented DNA fragments post-reaction. | SPRI/AMPure beads; critical for removing small mitochondrial fragments. |
| Transposase Enzyme | The core reagent that simultaneously fragments and tags accessible DNA. | Illumina Nextera Tn5, or custom-loaded Tn5. |
| qPCR Master Mix | Quantifies library yield and complexity prior to deep sequencing. | SYBR Green-based mixes with high-fidelity polymerase. |
Visualizing the Pre-Protocol Planning Workflow
Title: ATAC-seq Pre-Protocol Planning Decision Workflow
Title: Sample Preparation & Quality Control Pathway
Within the broader context of a thesis detailing the ATAC-seq (Assay for Transposase-Accessible Chromatin using sequencing) protocol, meticulous preparation is the cornerstone of success. This technical guide provides an exhaustive checklist of equipment and reagents, alongside core methodologies and data, essential for executing robust and reproducible ATAC-seq experiments. This pre-work ensures researchers, scientists, and drug development professionals can navigate the critical initial steps with confidence.
Core Equipment Checklist
A summary of essential instrumentation.
Table 1: Essential Laboratory Equipment for ATAC-seq
| Equipment Category | Specific Instrument/Item | Critical Function |
|---|---|---|
| Cell Processing | Cell culture hood (Biosafety Cabinet), CO2 incubator, centrifuge (refrigerated capable of 300-1000 RCF), hemocytometer or automated cell counter, water bath or heat block (37°C). | Aseptic cell handling, counting, and initial processing. |
| Nuclei Isolation & Transposition | Microcentrifuge, vortex mixer, pipettes (P2, P20, P200, P1000), low-retention microcentrifuge tubes (0.2 mL, 0.5 mL, 1.5 mL). | Precise reagent handling and nuclei preparation. |
| DNA Purification | Magnetic separation rack for DNA purification, thermomixer or incubator (37°C), Qubit fluorometer or equivalent. | Cleanup of transposed DNA and accurate quantification. |
| Library Preparation | Thermocycler (PCR machine), Agilent TapeStation, Bioanalyzer, or Fragment Analyzer. | Library amplification and quality assessment. |
| Sequencing | Illumina or other next-generation sequencing platform (typically off-site core facility). | High-throughput sequencing of final libraries. |
Reagent Checklist & The Scientist's Toolkit
Detailed list of consumables and critical reagent solutions.
Table 2: The ATAC-seq Scientist's Toolkit: Essential Reagents and Materials
| Reagent/Material | Function & Rationale | Example/Notes |
|---|---|---|
| Nuclei Isolation Buffer | Lyses the cell membrane while leaving the nuclear membrane intact, preserving chromatin state. | Typically contains Digitonin or NP-40, Tris-HCl, NaCl, MgCl2, Sucrose. |
| Tn5 Transposase | Enzyme complex that simultaneously fragments accessible DNA and adds sequencing adapters. | Commercially available as a loaded, active complex (e.g., Illumina Nextera Tn5). |
| Transposition Reaction Buffer | Provides optimal ionic and chemical conditions for Tn5 transposition activity. | Often supplied with the Tn5 enzyme; contains Mg2+. |
| DNA Purification Beads | SPRI (Solid Phase Reversible Immobilization) beads for size selection and cleanup of DNA. | AMPure XP beads or equivalent. Critical for removing reaction components and selecting fragments. |
| Library Amplification Reagents | PCR master mix, unique dual-indexed primers (i7 & i5). | Amplifies transposed DNA fragments and adds full-length sequencing adapters/indexes for multiplexing. |
| DNA Elution Buffer | Low-EDTA TE buffer or nuclease-free water for eluting purified DNA. | 10 mM Tris-HCl, pH 8.0 is standard. |
| Quality Control Reagents | DNA high-sensitivity assay kits (Qubit dsDNA HS), library quantification kits (qPCR-based). | Accurate quantification of low-concentration DNA pre- and post-amplification. |
| Viable Single-Cell Suspension | High-quality starting material. | >95% viability, 50,000-100,000 cells per reaction as a starting point. Avoid freeze-thaw cycles. |
Detailed Experimental Protocol: Nuclei Isolation & Tagmentation
Methodology: Nuclei Preparation from Cultured Cells
- Cell Harvest & Wash: Pellet 50,000-100,000 viable cells by centrifugation at 300-500 RCF for 5 minutes at 4°C. Aspirate supernatant completely.
- Cold Lysis: Resuspend cell pellet in 50 μL of cold ATAC-seq Lysis Buffer (10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl2, 0.1% IGEPAL CA-630 or 0.01% Digitonin). Vortex immediately for 5 seconds.
- Nuclei Pellet: Centrifuge at 500 RCF for 10 minutes at 4°C. Carefully aspirate supernatant without disturbing the nuclei pellet.
- Wash: Gently resuspend the pellet in 50 μL of cold ATAC-seq Wash Buffer (10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl2, no detergent). Centrifuge again at 500 RCF for 10 minutes at 4°C. Aspirate supernatant completely.
- Tagmentation: Resuspend the purified nuclei pellet in 25 μL of Transposition Mix (12.5 μL 2x TD Buffer, 2.5 μL Tn5 Transposase, 10 μL nuclease-free water). Mix gently by pipetting.
- Incubate: Incubate the reaction at 37°C for 30 minutes in a thermomixer with mixing (1000 rpm). Immediately proceed to DNA purification.
Data Presentation: Typical QC Metrics
Table 3: Expected Quantitative Outcomes for Key ATAC-seq Steps
| Experimental Stage | Measurement | Target/Expected Range | Purpose of QC |
|---|---|---|---|
| Post-Nuclei Prep | Nuclei count & integrity | >80% intact by microscopy | Ensure sufficient intact nuclei for tagmentation. |
| Post-Tagmentation/Purification | DNA Concentration (Qubit HS) | 0.5 - 5 ng/μL in 10-20 μL | Confirm successful tagmentation and recovery. |
| Post-PCR Amplification | Library Concentration (Qubit HS) | 10 - 50 ng/μL | Confirm successful amplification. |
| Final Library QC | Fragment Size Distribution (TapeStation) | Peak ~150-300 bp (nucleosomal ladder pattern) | Validate periodicity indicative of successful ATAC-seq. No adapter dimer peak (~80 bp). |
| Final Library QC | Molarity (qPCR) | ≥ 2 nM for sequencing | Accurate loading onto sequencer. |
Visualization of Workflows
ATAC-seq Core Experimental Workflow
Tn5 Tagmentation Molecular Mechanism
Executing the ATAC-seq Protocol: A Detailed Step-by-Step Laboratory Guide
Within the stepwise execution of the Assay for Transposase-Accessible Chromatin with high-throughput sequencing (ATAC-seq), the initial phase of cell harvesting and lysis is critically determinative. This step aims to isolate a population of intact, high-quality nuclei, free from cytoplasmic contaminants, to ensure efficient and unbiased tagmentation by the Tn5 transposase. Compromised nuclear integrity or residual cellular debris can lead to aberrant tagmentation, high mitochondrial DNA contamination, and ultimately, poor-quality sequencing data. This guide details the technical considerations and protocols for this foundational step.
Critical Parameters and Quantitative Benchmarks
Successful nuclei isolation balances complete lysis of the plasma membrane with preservation of the nuclear envelope. Key variables include cell type, cell number, lysis buffer composition, detergent concentration, incubation time, and physical handling.
Table 1: Quantitative Benchmarks for Nuclei Isolation in ATAC-seq
| Parameter | Optimal Range | Impact of Deviation |
|---|---|---|
| Starting Cell Number | 50,000 - 100,000 (fresh) | Low: Poor library complexity. High: Nuclei aggregation, inefficient tagmentation. |
| Lysis Buffer Salt (e.g., KCl) | 10-50 mM | High: Can destabilize nuclei, cause clumping. Low: May reduce lysis efficiency. |
| Detergent (e.g., NP-40, Igepal CA-630) | 0.1% - 0.5% (v/v) | High: Ruptures nuclear membrane, releases genomic DNA. Low: Incomplete lysis, cytoplasmic contamination. |
| Lysis Incubation | 2-10 minutes (on ice) | Long: Nuclei degradation. Short: Incomplete lysis. |
| Nuclei Yield Post-Wash | 70-90% of input cells | Low: Indicates excessive loss from harsh lysis or centrifugation. |
| Nuclei Integrity (Microscopy) | >95% intact, smooth membrane | Low: Leads to high background & mitochondrial reads. |
Table 2: Common Cell Type-Specific Adjustments
| Cell/Tissue Type | Key Challenge | Recommended Modification |
|---|---|---|
| Adherent Cells | Enzyme-based harvesting can damage nuclei. | Use gentle cell dissociation buffer, not trypsin. Scrape on ice in cold PBS. |
| Blood Cells (PBMCs) | Red blood cell (RBC) contamination. | Include RBC lysis step (e.g., ACK buffer) prior to nuclear lysis. |
| Tissues | Hard to dissociate, heterogeneous. | Use mechanical homogenization (Dounce) followed by filtration (40-70 µm). |
| Neurons / Fibroblasts | Robust cytoskeleton, hard to lyse. | Consider slightly higher detergent (0.2%) or brief (30 sec) room temp lysis. |
Detailed Experimental Protocol: Nuclei Isolation from Cultured Cells
This protocol is adapted from the Omni-ATAC method and current best practices for a wide range of mammalian cell lines.
Reagents & Equipment
- Cold Phosphate-Buffered Saline (PBS)
- Cell Lysis Buffer (freshly prepared on ice): 10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl₂, 0.1% Igepal CA-630, 0.1% Tween-20, 0.01% Digitonin (optional, enhances lysis for some cells).
- Nuclei Wash Buffer: 10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl₂, 0.1% Tween-20.
- 1% (w/v) Bovine Serum Albumin (BSA) in PBS or Nuclei Wash Buffer.
- Refrigerated centrifuge, swing-bucket rotor preferred.
- Low-retention microcentrifuge tubes (1.5 mL or 2 mL).
- Hemocytometer or automated cell counter.
- Fluorescence microscope with DNA stain (e.g., DAPI, Hoechst).
Procedure
- Cell Harvesting: For adherent cells, gently scrape culture dish on ice using cold PBS. For suspension cells, pellet directly. Centrifuge at 500 x g for 5 minutes at 4°C. Discard supernatant completely.
- Cell Counting: Resuspend cell pellet in 1 mL cold PBS. Take an aliquot, mix with trypan blue, and count viable cells. Centrifuge again at 500 x g for 5 minutes at 4°C.
- Plasma Membrane Lysis: Resuspend the cell pellet (50,000-100,000 cells) in 50 µL of cold Lysis Buffer. Vortex briefly at low speed or pipette mix gently. Incubate on ice for 3-5 minutes. Monitor lysis under a microscope: intact cells should disappear, leaving round, refractive nuclei.
- Washing: Immediately add 1 mL of cold Nuclei Wash Buffer to dilute the detergent. Invert tube gently 3-5 times to mix.
- Pellet Nuclei: Centrifuge at 500 x g for 10 minutes at 4°C. Carefully decant supernatant. The nuclei pellet may be loose and translucent.
- Wash (Optional but Recommended): Gently resuspend the pellet in 1 mL of cold Nuclei Wash Buffer with 1% BSA. Centrifuge again at 500 x g for 10 minutes at 4°C. BSA helps reduce nuclei sticking to tubes.
- Resuspension and QC: Discard supernatant. Gently resuspend the purified nuclei in an appropriate volume (e.g., 50 µL) of tagmentation buffer or PBS with 1% BSA. Count nuclei using a hemocytometer stained with DAPI (1:1000). Assess integrity (>95% intact) and absence of cellular debris. Proceed immediately to tagmentation.
The Scientist's Toolkit: Essential Reagents for Cell Lysis & Nuclei Isolation
Table 3: Key Research Reagent Solutions
| Reagent | Function/Principle | Critical Note |
|---|---|---|
| Igepal CA-630 (Nonidet P-40) | Non-ionic detergent. Disrupts lipid bilayers (plasma membrane) while sparing nuclear membranes at low concentrations. | Preferred over NP-40 for consistency. Concentration is critical (typically 0.1%). |
| Digitonin | Mild, cholesterol-specific detergent. Enhances plasma membrane permeabilization without damaging nuclei. | Used as a supplement (0.01%) in "Omni-ATAC" for difficult-to-lyse cells. |
| Tween-20 | Non-ionic detergent, milder than Igepal. Used in wash buffers to prevent nuclei clumping without causing further lysis. | Replaces Igepal in wash steps to maintain nuclear integrity. |
| Magnesium (Mg²⁺) Divalent Cations | Stabilizes chromatin and nuclear structure. Essential component of lysis and wash buffers. | Omission leads to nuclear swelling and rupture. Typical concentration is 3 mM. |
| Bovine Serum Albumin (BSA) | Acts as a blocking agent, reducing non-specific binding of transposase or nuclei to tube walls. | Inclusion in final resuspension buffer improves nuclei recovery and tagmentation uniformity. |
| Sucrose or Glycerol | Osmolyte. Can be added to buffers (e.g., 10% sucrose) to provide osmotic support, protecting nuclei from shear stress. | Particularly useful for sensitive primary cells or long-term nuclei storage. |
Workflow and Pathway Visualization
Title: ATAC-seq Nuclei Isolation Workflow and Buffer Components
Title: Impact of Lysis Quality on ATAC-seq Data Outcomes
Within the broader thesis on the ATAC-seq protocol, the tagmentation reaction is the pivotal enzymatic step that determines library complexity, insert size distribution, and overall data quality. This step utilizes a hyperactive Tn5 transposase pre-loaded with sequencing adapters to simultaneously fragment chromatin and tag the resulting DNA fragments with adapter sequences. This technical guide details the core parameters governing this reaction.
Core Reaction Parameters & Optimization
The efficiency and outcome of tagmentation are controlled by several interdependent variables. Optimal conditions balance sufficient fragmentation for resolution with the preservation of long fragments for nucleosome positioning analysis.
Table 1: Core Quantitative Parameters for Tn5 Tagmentation Optimization
| Parameter | Typical Range | Impact on Outcome | Optimal Starting Point for ATAC-seq |
|---|---|---|---|
| Temperature | 37°C - 55°C | Higher temperatures increase activity but risk enzyme denaturation and damaging epitopes. | 37°C |
| Incubation Time | 5 min - 60 min | Longer time increases fragment count but reduces median insert size. Critical for nuclei. | 30 min (for permeabilized nuclei) |
| Transposase Amount | 2.5 - 100 ng | Higher amounts increase fragmentation; requires titration to match cell count. | ~50,000 nuclei: 2.5-5 µL of commercial enzyme mix |
| Cell/Nuclei Count | 500 - 100,000 cells | Too high causes under-tagmentation; too low leads to over-fragmentation and PCR duplicate bias. | 50,000 viable nuclei |
| Mg²⁺ Concentration | 1 - 10 mM | Essential cofactor. Concentration directly drives transposition rate. | As supplied in buffer (typically ~10 mM final) |
| Reaction Volume | 10 - 50 µL | Affects effective concentration of all components. Consistency is key. | 25 µL (scalable) |
Table 2: Effect of Variable Manipulation on Final Library Metrics
| Altered Parameter | Direction of Change | Effect on Insert Size | Effect on Library Complexity | Risk if Suboptimal |
|---|---|---|---|---|
| Incubation Time | Increase | Decreases | Increases initially, then plateaus | Over-fragmentation (<100 bp fragments) |
| Enzyme Amount | Increase | Decreases | Increases | Loss of nucleosomal signal; adapter dimers |
| Cell/Nuclei Input | Increase | Increases | Increases (to a point) | Under-tagmentation; low unique read yield |
| Mg²⁺ Concentration | Increase | Decreases | Increases | Non-specific fragmentation activity |
Detailed Experimental Protocol: Tagmentation of Permeabilized Nuclei
This protocol assumes nuclei have been isolated, counted, and pelleted.
- Reagent Preparation: Thaw all components (Tagmentation Buffer, Tn5 Transposase) on ice. Prepare a master mix for multiple reactions to minimize variability.
- Master Mix Assembly (for 1 reaction):
- Nuclease-free H₂O: to 25 µL final volume.
- Tagmentation Buffer (2X): 12.5 µL.
- Tn5 Transposase: 2.5 µL (commercial pre-loaded enzyme, e.g., from Illumina).
- Mix gently by pipetting. Do not vortex.
- Reaction Assembly: Resuspend the pelleted nuclei (50,000 cells) directly in 25 µL of the master mix. Mix gently by pipetting up and down 5-10 times.
- Incubation: Incubate the reaction at 37°C for 30 minutes in a thermal cycler with heated lid (set to ≥70°C).
- Cleanup: Immediately add 250 µL of DNA Binding Buffer (from a MinElute or similar PCR cleanup kit) to the reaction. Mix thoroughly. Proceed to column-based purification per kit instructions, or add EDTA to 10 mM to chelate Mg²⁺ and halt the reaction if pausing.
Visualization of Workflow and Logical Decision Points
Title: ATAC-seq Tagmentation Optimization Workflow
Title: Tn5 Transposase Molecular Mechanism in Tagmentation
The Scientist's Toolkit: Key Reagent Solutions
Table 3: Essential Research Reagents for the Tagmentation Reaction
| Reagent / Material | Function & Rationale | Example (Commercial) |
|---|---|---|
| Hyperactive Tn5 Transposase (Pre-loaded) | Engineered enzyme for high activity at 37°C. Pre-loaded with sequencing adapters enables "one-pot" reaction. | Illumina Nextera Tn5, ThruPLEX Tagmentase. |
| Tagmentation Buffer (with Mg²⁺) | Provides optimal ionic strength and pH. Contains Mg²⁺, the essential divalent cation cofactor for transposition catalysis. | Often supplied with enzyme (e.g., TD Buffer from Illumina). |
| Digitonin or NP-40 | Detergent used in nuclei isolation and/or tagmentation buffer to permeabilize nuclear membranes, allowing Tn5 access. | Research-grade, low-concentration (e.g., 0.01%-0.1%). |
| PCR Clean-up Kit (SPRI Beads) | For immediate post-tagmentation purification to remove salts, enzyme, and stop the reaction. Critical for PCR step. | AMPure XP, MinElute PCR Purification Kit. |
| EDTA (0.5 M, pH 8.0) | Mg²⁺ chelator. An immediate stop solution if a column cleanup cannot be performed immediately post-incubation. | Molecular biology grade stock solution. |
| Nuclease-free Water | Used in master mix and elution. Essential to prevent non-specific degradation of DNA and adapters. | Certified, DEPC-treated, or ultrapure filtered. |
| Qubit dsDNA HS Assay Kit | Fluorometric quantitation of tagmented DNA pre-PCR. More accurate than absorbance for low-concentration, adapter-ligated DNA. | Thermo Fisher Scientific Qubit kit. |
| TapeStation/Bioanalyzer | Capillary electrophoresis system for QC of final library insert size distribution post-PCR. Assesses nucleosomal ladder pattern. | Agilent High Sensitivity DNA kit. |
Within the systematic framework of an ATAC-seq protocol step-by-step explanation, Step 3 is a critical juncture that bridges tagmentation and sequencing. This phase consists of two integrated procedures: the purification of tagmented DNA and the subsequent amplification of this material to create a sequencing-ready library. The primary objectives are to remove enzyme complexes and buffer components, to selectively enrich for properly tagmented fragments, and to append full sequencing adapters with sample-specific indices.
Post-Tagmentation Cleanup
Immediately following tagmentation, the reaction must be cleaned to halt Tn5 activity and to prepare the DNA for PCR. A common method employs a DNA purification kit utilizing silica-membrane columns or SPRI (Solid Phase Reversible Immobilization) bead-based cleanup.
Detailed Protocol: SPRI Bead Cleanup
- Add Binding Buffer: Combine the tagmentation reaction (typically 20 µL) with 20 µL of nuclease-free water and 40 µL of well-resuspended SPRI beads (at a 1:1 beads-to-sample ratio) in a low-binding microcentrifuge tube. Mix thoroughly by pipetting.
- Incubate: Room temperature incubation for 5 minutes.
- Pellet Beads: Place the tube on a magnetic stand until the supernatant is clear (~2-5 minutes). Carefully remove and discard the supernatant.
- Wash: With the tube on the magnet, add 200 µL of freshly prepared 80% ethanol without disturbing the bead pellet. Incubate for 30 seconds, then remove and discard the ethanol. Repeat this wash a second time.
- Dry: Briefly air-dry the bead pellet for ~1-3 minutes until it appears matte, ensuring no residual ethanol remains.
- Elute: Remove the tube from the magnet. Resuspend the dried beads in 21 µL of nuclease-free water or low-EDTA TE buffer. Incubate at room temperature for 2 minutes.
- Recover DNA: Place the tube back on the magnetic stand until the supernatant is clear. Transfer 20 µL of the purified eluate containing tagmented DNA to a new tube for PCR.
Library Amplification via PCR
The purified DNA is then amplified by PCR. This step serves to: 1) Enrich for fragments that have adapters ligated to both ends, 2) Attach full-length sequencing adapters and dual-index barcodes for sample multiplexing, and 3) Generate sufficient quantity for sequencing.
Detailed Protocol: PCR Amplification
- Assemble Reaction: Combine the following components in a PCR tube:
- 20 µL Purified tagmented DNA
- 2.5 µL Forward PCR Primer (i5 index, 10 µM)
- 2.5 µL Reverse PCR Primer (i7 index, 10 µM)
- 25 µL 2x High-Fidelity PCR Master Mix (e.g., NEB Next High-Fidelity 2x MM)
- Thermocycling: Perform amplification using the following conditions:
- 72°C for 5 minutes (gap filling)
- 98°C for 30 seconds (initial denaturation)
- Cycle n times:
- 98°C for 10 seconds (denaturation)
- 63°C for 30 seconds (annealing/extension)
- Hold at 4°C.
- Cycle Number Determination: The optimal number of PCR cycles (n) is determined by a preliminary qPCR side-reaction to avoid over-amplification. A 5 µL aliquot of the cleanup eluate is amplified with SYBR Green and the same primers. The cycle number corresponding to ¼–⅓ of the maximum fluorescence is chosen for the main library PCR.
Table 1: Key Quantitative Parameters for Step 3
| Parameter | Typical Value/Range | Purpose/Note |
|---|---|---|
| SPRI Bead Ratio | 1.0x (Sample Volume) | Binds fragments > ~100 bp; removes primers, buffers, and small fragments. |
| Post-Cleanup Elution Volume | 20-22 µL | Maximizes DNA recovery for PCR input. |
| PCR Input DNA Volume | 20 µL (entire eluate) | Uses all recovered material due to low yield. |
| PCR Cycle Number (n) | 8-12 cycles | Must be determined empirically via qPCR to prevent GC/sequence bias. |
| Final Library Concentration Target | > 5 nM (Qubit/qPCR) | Ensures sufficient material for sequencing cluster generation. |
| Optimal Library Size Distribution | 150-800 bp peak (Bioanalyzer/TapeStation) | Mononucleosomal (~200 bp) and dinucleosomal (~400 bp) fragments. |
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Post-Tagmentation and Amplification
| Item | Function & Rationale |
|---|---|
| SPRI Magnetic Beads | Selective binding and purification of DNA fragments based on size; removes salts, enzymes, and short fragments. |
| 80% Ethanol (freshly prepared) | Wash buffer to remove salts and impurities from bead-bound DNA without causing elution. |
| Nuclease-free Water or Low-EDTA TE | Elution buffer; low EDTA prevents interference with subsequent enzymatic steps. |
| High-Fidelity PCR Master Mix | Provides thermostable polymerase, dNTPs, Mg2+, and optimized buffer for efficient, low-bias amplification. |
| Dual-Indexed PCR Primers (i5 & i7) | Contain full P5/P7 flow cell binding sites, sample-specific barcodes, and sequences complementary to the Nextera Transposon end. |
| SYBR Green qPCR Master Mix | For real-time monitoring of library amplification to determine the optimal, non-saturating cycle number. |
| Magnetic Stand | For separation of SPRI beads from solution during cleanup steps. |
| Low-Binding Microcentrifuge Tubes | Minimizes DNA loss through surface adhesion. |
Visualization of Step 3 Workflow and Logic
Title: ATAC-seq Step 3: Cleanup & Amplification Workflow
Title: PCR Completes Sequencing Adapters
Within the broader ATAC-seq protocol, Step 4 is the critical juncture where the transposed and amplified DNA library is prepared for the sequencer. This step ensures the removal of enzymatic reagents, PCR primers, and small fragments, ultimately yielding a library of the correct size distribution, purity, and concentration for high-quality sequencing data. Failure in proper purification, Quality Control (QC), and quantification is a primary source of experimental failure in ATAC-seq workflows.
Library Purification: Methodologies and Rationale
Post-PCR amplification, the reaction mixture contains the target library fragments, excess primers, primer dimers, nucleotides, salts, and enzymes. Purification serves to isolate fragments within the desired size range (typically 100-700 bp for ATAC-seq, representing mononucleosomal and multinucleosomal fragments).
1. Solid-Phase Reversible Immobilization (SPRI) Bead Clean-up This is the most widely adopted method due to its speed, efficiency, and ability to perform size selection.
- Principle: Paramagnetic beads coated with carboxyl groups bind DNA in the presence of a high concentration of PEG and salt. The binding affinity is size-dependent, allowing for selective isolation of fragments above a threshold.
- Detailed Protocol:
- Allow AMPure XP or SPRIselect beads to reach room temperature.
- Vortex beads to ensure a homogeneous suspension.
- Combine the PCR reaction with beads at a defined sample-to-bead ratio (e.g., 0.5x to 1.8x). A 0.5x ratio is often used to remove large fragments and primer dimers (<100 bp), while a 1.0-1.8x double-sided clean-up (sequentially removing both small and large fragments) is used for strict size selection.
- Incubate at room temperature for 5 minutes.
- Place the tube on a magnetic stand until the supernatant is clear (~5 minutes).
- Carefully remove and discard the supernatant.
- With the tube on the magnet, wash the bead-bound DNA twice with 200 µL of freshly prepared 80% ethanol. Incubate for 30 seconds per wash before removing.
- Air-dry the beads for 5-10 minutes until cracks appear. Do not over-dry.
- Elute DNA in nuclease-free water or TE buffer (e.g., 20-30 µL) by pipetting. Incubate for 2 minutes off the magnet.
- Place back on the magnet, and transfer the eluted library to a new tube.
2. Gel Electrophoresis-Based Size Selection Considered the "gold standard" for precise size selection but is more labor-intensive and lower throughput.
- Principle: The library is run on a high-resolution gel (e.g., 2% agarose or Pippin Prep gel cassette), and fragments within a specific window are excised and purified.
- Detailed Protocol:
- Prepare a 2% agarose gel in 1x TAE with a DNA-safe stain (e.g., SYBR Safe).
- Load the amplified library alongside an appropriate DNA ladder.
- Run the gel at low voltage (e.g., 80-100 V) for optimal resolution.
- Visualize the library smear under blue light. Using a clean scalpel, excise the gel slice corresponding to the target size range (e.g., 150-500 bp).
- Purify DNA from the gel slice using a commercially available gel extraction kit (e.g., QIAquick Gel Extraction Kit), following the manufacturer's instructions for binding, washing, and elution.
Quality Control (QC) Assessment
QC validates the success of purification and assesses library integrity prior to sequencing.
1. Fragment Size Distribution Analysis (Bioanalyzer/TapeStation) This is the most informative QC step for ATAC-seq libraries.
- Method: Uses microfluidic capillary electrophoresis to provide an electrophoretogram and pseudo-gel image.
- Expected Output: A smooth, periodic distribution of fragment sizes with peaks approximately every 200 bp (reflecting nucleosomal periodicity: mononucleosome ~200 bp, dinucleosome ~400 bp, etc.). The absence of a peak at ~100 bp indicates successful removal of adapter dimers. A representative size profile is shown in Table 1.
2. Library Concentration and Purity (Fluorometry & Spectrophotometry)
- Qubit Fluorometer: Uses dsDNA High Sensitivity (HS) assay. Provides highly accurate concentration measurements without interference from RNA or free nucleotides.
- NanoDrop Spectrophotometer: Assesses purity via A260/A280 (~1.8 for pure DNA) and A260/A230 (>2.0) ratios. Can indicate contamination from proteins, phenols, or salts. Less accurate for low-concentration samples.
Library Quantification for Sequencing
Accurate molarity determination is essential for optimal cluster density on the flow cell.
1. Quantitative PCR (qPCR) The most accurate method for quantifying amplifiable library molecules. It mirrors the bridge amplification process of Illumina sequencers and is not fooled by adapter dimers or contaminating genomic DNA.
- Protocol (KAPA Library Quantification Kit):
- Perform a 1:10,000 to 1:1,000,000 dilution of the purified library.
- Prepare serial dilutions of the provided DNA standard.
- Prepare a master mix containing SYBR Green, primers specific to the Illumina adapter sequences, and water.
- Combine master mix with standards and diluted library samples in a qPCR plate.
- Run the qPCR program (e.g., 95°C for 5 min, then 35 cycles of 95°C for 30s and 60°C for 45s).
- Generate a standard curve from the standards and calculate the amplifiable concentration (nM) of the library sample.
Data Presentation
Table 1: QC Metrics for a Successful ATAC-seq Library
| QC Method | Target Metric | Optimal Result | Indication of Problem |
|---|---|---|---|
| Bioanalyzer HS DNA | Peak Size Distribution | Major peak ~200-300 bp, periodicity to ~700 bp | Large peak at <150 bp (adapter dimer) or >1000 bp (over-transposition/incomplete purification) |
| Qubit dsDNA HS | Concentration | > 1 ng/µL in elution volume | Very low yield may indicate poor transposition or PCR amplification |
| NanoDrop | A260/A280 | 1.8 - 2.0 | Ratio <1.7 suggests protein/phenol contamination |
| NanoDrop | A260/A230 | 2.0 - 2.2 | Ratio <1.8 suggests salt/carbohydrate contamination |
| qPCR (KAPA) | Amplifiable Concentration | Typically 2 - 20 nM | Large discrepancy vs. Qubit suggests high adapter-dimer content |
Table 2: The Scientist's Toolkit: Essential Reagents for ATAC-seq Library Clean-up & QC
| Item | Function/Description | Example Product |
|---|---|---|
| SPRI Beads | Size-selective purification of DNA fragments; removes primers, dimers, and salts. | AMPure XP, SPRIselect |
| Ethanol (80%) | Wash solution for SPRI bead clean-up; removes residual salts and impurities. | Freshly prepared in nuclease-free water |
| Nuclease-Free Water/TE Buffer | Elution buffer for purified DNA libraries. Stabilizes DNA for storage. | Invitrogen, Teknova |
| High Sensitivity DNA Assay Chips | Microfluidic chips for precise fragment analysis on Bioanalyzer. | Agilent High Sensitivity DNA Kit |
| DNA HS Screentapes | Pre-cast gels for automated fragment analysis on TapeStation. | Agilent D5000/High Sensitivity D1000 |
| Qubit dsDNA HS Assay Kit | Fluorometric dye for accurate quantification of low-concentration dsDNA. | Invitrogen Qubit dsDNA HS Assay |
| Library Quantification Kit | qPCR-based kit with adapter-specific primers to determine amplifiable molarity. | KAPA Library Quantification Kit, Illumina Library Quantification Kit |
| Size Selection Gel Cassettes | Automated, precise gel-based size selection system. | Sage Science Pippin Prep Cassettes |
Visualizations
ATAC-seq Library Prep: Purification to Sequencing Readiness
SPRI Bead Clean-up Workflow for ATAC-seq Libraries
The optimization of sequencing parameters is a critical, resource-intensive step in the ATAC-seq (Assay for Transposase-Accessible Chromatin using sequencing) workflow. Framed within a broader thesis on a step-by-step ATAC-seq protocol, this step directly impacts data quality, interpretability, and cost. This guide provides evidence-based recommendations for read depth, read type, and platform selection tailored for researchers, scientists, and drug development professionals.
Sequencing Read Depth: Balancing Sensitivity and Cost
Sequencing depth determines the power to detect open chromatin regions. Inadequate depth leads to poor peak calling; excessive depth yields diminishing returns. Recommendations are stratified by common experimental goals.
Table 1: Recommended Sequencing Depth for ATAC-seq Applications
| Experimental Goal | Minimum Recommended Depth per Sample (Passing Filter Reads) | Optimal Depth per Sample | Primary Rationale |
|---|---|---|---|
| Global Chromatin Accessibility Profiling (e.g., identifying major cell type differences) | 25-50 million reads | 50-75 million reads | Covers a high proportion of accessible sites in the genome with good reproducibility. |
| Differential Peak Analysis (Comparing conditions or cell states) | 50 million reads | 75-100 million reads | Enables robust statistical comparison and detection of subtle, condition-specific changes. |
| Transcription Factor Footprinting | 100 million reads | 200+ million reads | High depth is required to resolve the sparse, strand-specific cleavage patterns indicative of TF binding. |
| Single-Cell ATAC-seq (scATAC-seq) | Aggregate ~100-150 million reads across all cells | Aggregate ~200+ million reads across all cells | While per-cell depth is low (~5-50k reads), aggregate depth must be high to capture rare cell populations and their distinct accessibility profiles. |
Protocol Note: Estimating Depth Needs
- Pilot Experiment: For a new cell type or condition, sequence 2-3 libraries to 25M reads each. Perform peak calling at subsampled depths (e.g., 10M, 25M, 50M reads).
- Saturation Analysis: Plot the number of unique non-redundant fragments or called peaks against sequencing depth. The "elbow" where the curve plateaus indicates a sufficient depth.
- Replication > Depth: For differential analysis, allocating resources to 3-4 biological replicates at 50M reads each is generally more powerful than 1-2 replicates at 100M reads.
Paired-End vs. Single-End Sequencing
While early ATAC-seq used single-end (SE) sequencing, paired-end (PE) is now the standard for bulk ATAC-seq due to significant advantages.
Table 2: Paired-End vs. Single-End for ATAC-seq
| Aspect | Paired-End (PE) | Single-End (SE) | Recommendation |
|---|---|---|---|
| Fragment Size Distribution | Directly measurable. Enables precise nucleosome positioning analysis (mono-, di-, tri-nucleosome peaks). | Inferred indirectly, less accurate. | PE is mandatory for nucleosome occupancy/positioning studies. |
| Insertion Site Mapping | Higher precision in mapping the exact Tn5 integration site (accessible region). | More ambiguous mapping, especially for reads in repetitive regions. | PE strongly preferred for improved mapping accuracy and sensitivity. |
| Data Quality | Enables detection of PCR duplicates with higher confidence based on both coordinates of a fragment. | Duplicate marking is less accurate, potentially leading to over-removal of true signal. | PE is standard for optimal data processing. |
| Transcription Factor Footprinting | Superior for detecting the ~10 bp periodicity of Tn5 cleavage within a footprint. | Possible, but with reduced resolution and confidence. | PE is essential for serious footprinting analysis. |
| Cost | ~1.7-2x the cost of SE sequencing per sample. | Lower cost. | PE is strongly recommended for all bulk ATAC-seq. SE may be considered for cost-limited pilot/scaling studies where nucleosome data is not needed. |
Experimental Protocol: Library QC for PE Sequencing
- Quality Control: Prior to sequencing, validate library fragment size distribution using a Bioanalyzer or TapeStation. A successful ATAC-seq library shows a clear periodicity of fragments ~200 bp apart (nucleosomal ladder).
- Sequencing Configuration: For PE sequencing on Illumina platforms, a common configuration is PE 50 bp (or 75 bp) x 2. This read length is sufficient to map most fragments, given the sub-nucleosomal size selection (< 700 bp) during library prep. For footprinting, PE 100 bp x 2 or longer may be beneficial.
Platform Choice: Throughput, Read Length, and Cost
The Illumina platform dominates ATAC-seq due to its high accuracy and throughput, but new entrants are relevant for specific use cases.
Table 3: Sequencing Platform Comparison for ATAC-seq
| Platform (Vendor) | Optimal Use Case | Key Advantage | Consideration for ATAC-seq |
|---|---|---|---|
| NovaSeq X & 6000 (Illumina) | Large-scale projects: population studies, drug screening (100s-1000s of samples), deep footprinting. | Extremely high throughput, lowest cost per Gb. | Best for core facilities. Requires sample multiplexing to utilize full flow cell capacity cost-effectively. |
| NextSeq 1000/2000 (Illumina) | Mid-scale projects: differential analysis, multi-replicate experiments (10s-100s of samples). | Balance of throughput and flexibility. P2 flow cell enables high-output runs; P3 enables rapid, lower-output runs. | The workhorse for most academic labs. Ideal for generating 50-100M PE reads per sample across many samples. |
| MiSeq (Illumina) | Protocol optimization, pilot runs, and library QC. | Fast turnaround, long read lengths possible. | Low throughput. Useful for testing new cell types or conditions before scaling up. |
| Ultima Genomics | Exploratory studies requiring ultra-deep sequencing (e.g., footprinting in rare samples). | Very low cost per Gb. | Emerging technology; bioinformatic pipelines may require adaptation. Read length currently shorter than Illumina. |
| Element AVITI | Projects requiring long reads or specific cost structures. | Competitive cost, flexible read lengths. | Gaining traction; compatibility with standard ATAC-seq bioinformatics should be verified. |
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for ATAC-seq Library Sequencing
| Item | Function / Purpose | Example Product / Note |
|---|---|---|
| Indexed Sequencing Adapters | Enables multiplexing of multiple libraries on a single sequencing run. Unique dual indices (UDIs) are strongly recommended to reduce index hopping. | Illumina IDT for Illumina UD Indexes, Nextera XT Index Kit v2. |
| Library Quantification Kit | Accurate quantification of final library concentration is critical for pooling and loading equimolar amounts. | Qubit dsDNA HS Assay Kit, qPCR-based kits (e.g., KAPA Library Quantification Kit). |
| Size Selection Reagents | Optional post-amplification clean-up to remove primer dimers and select optimal fragment range. | SPRIselect beads (Beckman Coulter) used in a double-sided size selection. |
| High-Fidelity PCR Mix | Used during the library amplification step prior to sequencing. Critical for minimal bias. | NEBNext Ultra II Q5 Master Mix, KAPA HiFi HotStart ReadyMix. |
| Sequencing Control | Spike-in control to monitor sequencing run performance. | Illumina PhiX Control v3 (typically 1% of total load). |
| Sequencing Reagent Kits | Platform-specific flow cell and chemistry kits. | Illumina NovaSeq X Plus 25B Reagent Kit, NextSeq P2 200/300 cycle kits. |
Workflow and Decision Pathway Diagrams
Diagram 1 Title: ATAC-seq Sequencing Parameter Decision Pathway
Diagram 2 Title: From ATAC-seq Library Prep to Sequencing Data Files
ATAC-seq Troubleshooting: Solving Common Problems and Optimizing Your Data Quality
Within the context of a comprehensive ATAC-seq (Assay for Transposase-Accessible Chromatin using sequencing) protocol, library preparation failures represent a critical bottleneck. This technical guide provides a systematic diagnostic framework, from initial nuclei isolation to final PCR amplification, to troubleshoot poor yield or complete library failure.
Assessing Nuclei Quality: The Primary Determinant
The integrity of isolated nuclei is the foundational step in ATAC-seq. Compromised nuclei yield poor chromatin accessibility data and subsequent library failures.
Experimental Protocol: Nuclei Quality Assessment via Flow Cytometry & Microscopy
- Staining: Resuspend ~50,000 nuclei in 1x PBS containing 1 µg/mL DAPI (4',6-diamidino-2-phenylindole) or 0.5 µM SYTOX Green.
- Flow Cytometry: Analyze using a flow cytometer with a 405 nm (DAPI) or 488 nm (SYTOX) laser. Record events for 1 minute at a slow flow rate.
- Gating Strategy: Gate on singlet events based on forward scatter area vs. height, then plot fluorescence intensity. Intact nuclei show a tight, high-fluorescence population.
- Microscopy Validation: Image 10 µL of stained nuclei on a hemocytometer using a fluorescence microscope with appropriate filters. Assess morphology.
Table 1: Nuclei Quality Metrics and Implications
| Metric | Acceptable Range | Suboptimal Range | Implication for ATAC-seq |
|---|---|---|---|
| Viability (DAPI-/SYTOX-) | >85% | 70-85% | Reduced complexity, high mitochondrial reads. |
| Concentration | 5,000-10,000 nuclei/µL | <2,000 or >20,000 nuclei/µL | Under- or over-tagmentation, affecting fragment distribution. |
| Intact Morphology | >90% spherical, smooth | High debris, irregular shapes | Premature chromatin release, high background. |
| Aggregation | <5% clumped nuclei | >15% clumped nuclei | Inconsistent tagmentation, low yield. |
Title: Nuclei Quality Control Workflow for ATAC-seq
Tagmentation Optimization: Enzyme and Reaction Conditions
Inefficient transposase (Tn5) activity is a common failure point, leading to low library yield or skewed fragment sizes.
Experimental Protocol: Titrating Transposase Input
- Set up 50 µL tagmentation reactions with fixed nuclei count (e.g., 50,000) in provided buffer.
- Titrate the commercial Tn5 enzyme (e.g., 0.5x, 1x, 2x, 4x of the standard volume).
- Incubate at 37°C for 30 minutes with mild shaking (300 rpm).
- Immediately purify DNA using a silica-column based clean-up kit. Elute in 20 µL.
- Analyze 1 µL on a Bioanalyzer (High Sensitivity DNA chip) or TapeStation to visualize the fragment size distribution.
Table 2: Tagmentation Troubleshooting Guide
| Symptom | Bioanalyzer Profile | Potential Cause | Experimental Fix |
|---|---|---|---|
| No Fragments | No peak, only lower marker. | Inactive Tn5, Inhibitors in nuclei prep, Incorrect Mg²⁺ concentration. | Fresh Tn5 aliquot, Clean nuclei with wash steps, Verify buffer composition. |
| High Molecular Weight Smear | Large smear > 2,000 bp. | Insufficient Tn5, Short incubation time, Low temperature. | Increase Tn5 titration, Extend incubation to 45-60 min, Verify thermal cycler calibration. |
| Over-digestion | All fragments < 100 bp. | Excess Tn5, Excessive incubation time, Too many nuclei. | Reduce Tn5 amount, Reduce time to 15 min, Re-quantify nuclei input. |
| Bimodal Distribution | Peaks at ~200 bp and > 1,000 bp. | Nuclei clumping/aggregation, Incomplete reaction mixing. | Filter nuclei pre-reaction, Ensure gentle but thorough pipette mixing. |
Title: Tn5 Titration Impact on Fragment Profile
Library Amplification: PCR Pitfalls and QC
The final PCR step enriches tagmented DNA but introduces biases and errors if not optimized.
Experimental Protocol: qPCR-based Cycle Determination
- Set up a 25 µL qPCR reaction with 2x SYBR Green Master Mix, library-specific primers, and 2-5 µL of purified tagmented DNA.
- Run on a real-time PCR machine with cycling: 72°C/5min, 98°C/30s, then cycle (98°C/10s, 63°C/30s, 72°C/60s) with plate read.
- Plot fluorescence (Rn) vs. cycle number. The optimal cycle number (Cq) is the cycle where the amplification curve crosses the threshold (mid-linear phase), typically between 8-14 cycles.
- Perform the scaled-up library PCR using (Cq - 1) cycles.
Table 3: Post-Amplification Library QC Metrics
| QC Method | Passing Criteria | Indication of Failure | Corrective Action |
|---|---|---|---|
| Qubit dsDNA HS Assay | Yield: > 10 nM from 50k nuclei. | Yield < 1 nM. | Repeat qPCR cycle determination; check PCR reagents. |
| Bioanalyzer/TapeStation | Clear peak ~200-600 bp; No primer dimers (~100 bp). | Large primer dimer peak, No library peak, Broad smear. | Re-optimize PCR clean-up with size selection; redesign primers. |
| qPCR for Library Quant (Kapa) | [Library] within 2-fold of Qubit reading. | [Library] << Qubit reading (inhibitors present). | Re-purify library; dilute template in subsequent PCR. |
| Fragment Analyzer | Nuclear DNA peak present; Mitochondrial DNA < 50%. | Mitochondrial DNA > 70%. | Improve nuclei purity; use longer Tn5 incubation for nuclear access. |
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for ATAC-seq Troubleshooting
| Reagent/Material | Function & Rationale | Example Product/Catalog |
|---|---|---|
| Digitonin | Permeabilizes cell membranes while leaving nuclear membranes intact for clean nuclei isolation. | Millipore Sigma, D141-100MG. |
| DAPI / SYTOX Green | DNA-intercalating dyes for flow cytometric quantification of nuclei integrity and viability. | Thermo Fisher, D1306 / S7020. |
| Tagmentase (Tn5) | Engineered transposase that simultaneously fragments and adapters DNA. Critical for open chromatin capture. | Illumina Tagment DNA TDE1 (20034197). |
| SPRIselect Beads | Size-selective magnetic beads for post-tagmentation and post-PCR clean-up to remove small fragments and primers. | Beckman Coulter, B23318. |
| High-Sensitivity DNA Assay Kits | Accurate quantification of low-concentration DNA libraries pre-sequencing. | Agilent Bioanalyzer HS DNA kit (5067-4626). |
| KAPA Library Quantification Kit | qPCR-based absolute quantification of amplifiable library molecules for accurate sequencing pool normalization. | Roche, KK4824. |
| PCR Enhancer (e.g., DMSO, BSA) | Additives that can improve PCR efficiency and specificity when amplifying GC-rich or complex genomic regions. | Thermo Fisher, 10769010. |
Title: Decision Tree for ATAC-seq Library Failure
1. Introduction in the Context of ATAC-seq Research The Assay for Transposase-Accessible Chromatin using sequencing (ATAC-seq) is a cornerstone technique for probing chromatin accessibility. A persistent technical challenge in ATAC-seq is the over-representation of mitochondrial DNA (mtDNA) reads, which can constitute 20-80% or more of total sequencing reads, drastically reducing usable data yield and increasing sequencing costs. This contamination arises because the mitochondrial membrane is permeabilized alongside the nuclear envelope by standard detergents in the protocol, exposing the abundant, protein-free mitochondrial genome to the hyperactive Tn5 transposase. Within a broader thesis on ATAC-seq optimization, this guide details the causes of mtDNA contamination and provides in-depth, actionable strategies for its reduction using digitonin and nuclease treatment.
2. Causes of Mitochondrial DNA Contamination in ATAC-seq
| Cause | Mechanism | Typical Impact on mtDNA Reads |
|---|---|---|
| Non-selective Permeabilization | Use of ionic detergents (e.g., NP-40, Tween-20) lyses all cellular membranes, including mitochondria. | High (50-80%) |
| Abundance of mtDNA | Each cell contains hundreds to thousands of mtDNA copies vs. two nuclear DNA copies. | Inherently High |
| Lack of Chromatinization | mtDNA is not protected by nucleosomes, making it a prime substrate for Tn5. | High |
| Cell Type Variation | Cells with high metabolic activity (e.g., cardiomyocytes, hepatocytes) have higher mtDNA content. | Variable (20-90%) |
3. Core Reduction Strategies: Principles and Protocols
3.1. Selective Permeabilization with Digitonin Digitonin, a plant-derived glycoside, selectively permeabilizes cholesterol-rich membranes (like the plasma membrane) over cholesterol-poor ones (like the mitochondrial inner membrane). This allows Tn5 access to the nucleus while theoretically leaving mitochondria intact.
Detailed Protocol: Titrated Digitonin Wash
- Cell Preparation: Isolate 50,000-100,000 viable cells in cold PBS. Pellet (500 RCF, 5 min, 4°C).
- Hypotonic Lysis Buffer: Prepare ice-cold buffer (10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl₂, 0.1% IGEPAL CA-630, 0.1% Tween-20, 0.01% Digitonin). Note: Digitonin concentration is critical.
- Permeabilization: Resuspend cell pellet in 50 µL of Hypotonic Lysis Buffer. Incubate on ice for 3 minutes.
- Immediate Quenching: Add 1 mL of Wash Buffer (10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl₂, 0.1% Tween-20) to quench digitonin.
- Centrifugation: Pellet nuclei (500 RCF, 10 min, 4°C). Carefully remove supernatant.
- Tn5 Tagmentation: Proceed immediately with the standard ATAC-seq tagmentation reaction on the purified nuclei pellet.
Optimization Requirement: The optimal digitonin concentration (typically 0.01-0.1%) must be empirically determined for each cell type to balance nuclear access and mitochondrial integrity.
3.2. Enzymatic Depletion with mtDNA-Targeting Nuclease This post-permeabilization approach actively degrades accessible mtDNA using a nuclease that is excluded from the nucleus due to its intact membrane.
- Detailed Protocol: Pre-Tagmentation Nuclease Treatment
- Standard Permeabilization: Lyse cells using a standard ATAC-seq lysis buffer (e.g., containing IGEPAL CA-630 or Tween-20) to permeabilize all membranes. Pellet nuclei and mitochondria (500 RCF, 10 min, 4°C).
- Nuclease Reaction Setup: Resuspend the pellet in 1X CutSmart Buffer (or compatible nuclease buffer). Add Exonuclease III (Exo III) or Plasmid-Safe ATP-Dependent DNase to a final concentration of 5-20 U/µL.
- Incubation: Incubate at 37°C for 15-30 minutes.
- Enzyme Inactivation/Removal: Add EDTA to 10 mM to chelate Mg²⁺ and inactivate nucleases, OR perform two washes with Wash Buffer (see 3.1).
- Tn5 Tagmentation: Proceed with the standard ATAC-seq tagmentation reaction.
4. Comparative Data Analysis of Reduction Strategies
| Strategy | Principle | Typical mtDNA Reduction | Advantages | Disadvantages |
|---|---|---|---|---|
| Standard Detergent (NP-40/Tween) | General lysis | Baseline (Reference) | Simple, robust | Very high mtDNA contamination |
| Titrated Digitonin | Selective membrane permeabilization | 50-90% reduction | Maintains nuclear integrity, simple addition | Requires cell-type optimization, can reduce signal |
| Exonuclease III Treatment | Enzymatic digestion of exposed DNA | 70-95% reduction | Highly effective, works post-lysis | Risk of nuclear DNA damage if nuclear envelope is compromised, extra step |
| Combined (Digitonin + Exo III) | Selective lysis + enzymatic digestion | >95% reduction | Maximal depletion | Most complex protocol, cumulative risk of nuclear damage |
5. Visualized Workflows and Logical Framework
Title: ATAC-seq mtDNA Reduction Strategy Workflow
Title: Logical Relationship of mtDNA Causes & Solutions
6. The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent | Function in mtDNA Reduction | Key Consideration |
|---|---|---|
| Digitonin (High-Purity) | Selective permeabilization of plasma membrane. | Solubility is low; prepare fresh stock in DMSO or water with heating. Critical to titrate. |
| Exonuclease III (E. coli) | Degrades double-stranded DNA from 3' ends. Preferentially attacks protein-free mtDNA. | Must be used before tagmentation. Mg²⁺ is required for activity. |
| Plasmid-Safe ATP-Dependent DNase | Degrades linear dsDNA, sparing circular supercoiled DNA (like mtDNA in some states). | Requires ATP. Efficiency for mtDNA reduction in ATAC-seq is variable. |
| IGEPAL CA-630 / NP-40 | Non-ionic detergent for standard nuclear isolation. | Causes high mtDNA contamination. Serves as a negative control for optimization. |
| Sucrose-Containing Wash Buffers | Maintains isotonicity to prevent organelle rupture during washes. | Helps preserve mitochondrial integrity when used with digitonin. |
| Dual-Lysis Buffers | Commercial kits providing separate plasma membrane and nuclear lysis buffers. | Often incorporate digitonin or saponin in the first lysis step. |
Optimizing Transposition Time and Transposase Concentration for Your Cell Type
Within the step-by-step execution of the Assay for Transposase-Accessible Chromatin using sequencing (ATAC-seq), the transposition reaction is the pivotal step that determines data quality. This step, where the Tn5 transposase simultaneously fragments and tags accessible genomic regions, is governed by two critical parameters: transposition time and transposase concentration. Optimizing these variables for your specific cell type—whether primary, cultured, or tissue-derived—is essential for generating libraries with optimal fragment size distribution, high signal-to-noise ratio, and minimal mitochondrial background. This guide provides a technical framework for systematic optimization, integral to a robust ATAC-seq thesis research project.
Core Principles and Rationale for Optimization
The Tn5 transposase operates by inserting adapter sequences into open chromatin regions. Excessive transposase or prolonged incubation can lead to over-fragmentation, increased background from closed chromatin, and elevated mitochondrial DNA reads (due to accessible mitochondrial genomes). Insufficient transposase or short incubation yields low library complexity and poor coverage. The optimal balance is cell-type-dependent due to variances in nuclear size, chromatin accessibility landscapes, and the presence of inhibitors.
The following table synthesizes optimization findings from recent studies across diverse cell types.
Table 1: Empirical Optimization Ranges for Different Cell Types
| Cell Type Category | Recommended Transposase Concentration (in 50 µL reaction) | Recommended Transposition Time | Key Rationale / Effect | Primary Citation Context (Recent Findings) |
|---|---|---|---|---|
| Fresh Primary Cells (e.g., PBMCs, neurons) | 2.5 – 5 µL of commercial enzyme (e.g., Illumina) | 30 min | Higher concentrations often needed for dense chromatin; time minimized to reduce mitochondrial artifact. | Omni-ATAC protocol adjustments (2021-2023) suggest 5 µL for difficult nuclei. |
| Cultured Cell Lines (e.g., HEK293, K562) | 2.5 µL of commercial enzyme | 30 min | Standard condition works well for most immortalized lines with consistent nuclei prep. | Benchmarking studies (2022) show 2.5µL/30min optimal for complexity vs. background in common lines. |
| Fresh/Frozen Tissue (e.g., liver, tumor biopsies) | 5 – 7.5 µL | 30 – 45 min | Increased enzyme and time to penetrate partially compacted nuclei from tissue dissociation. | Live Search Update: Modified ATAC (mATAC) for tissue (2024) recommends titration up to 7.5µL. |
| Low-Input Cells (< 10,000 nuclei) | 2.5 µL | 60 min | Extended time maximizes tagmentation efficiency from limited material. | Low-cell-number protocols (2023) favor longer incubation over more enzyme to conserve reagent. |
| Fixed Cells or Nuclei | 5 – 10 µL | 60 – 120 min | Chromatin cross-linking impedes Tn5; requires drastic increase in both parameters. | SHARE-ATAC & fixation-compatible methods (2023-2024) emphasize extensive optimization. |
Table 2: Outcome Metrics for Optimization Assessment
| Parameter to Measure | Optimal Outcome | Suboptimal (Too Low) | Suboptimal (Too High) | Assay |
|---|---|---|---|---|
| Post-PCR Library Size Distribution (Bioanalyzer) | Major peak ~200-600 bp (nucleosomal ladder). | Peak >1000 bp (under-fragmentation). | Smear <150 bp (over-fragmentation). | Bioanalyzer/TapeStation |
| Mitochondrial Read Percentage | < 20% (ideally <10%) for most cells. | Not typically affected. | Can exceed 50%. | Sequencing Analysis |
| Fraction of Reads in Peaks (FRiP) | > 20-30% for strong signal. | Low (<15%). | May decrease due to background. | Sequencing Analysis |
| Library Complexity (Unique Fragments) | High, saturating at reasonable sequencing depth. | Low. | May decrease due to PCR duplication. | Sequencing Analysis |
Detailed Experimental Protocol for Systematic Optimization
This protocol assumes nuclei have been successfully isolated and purified from your target cell type.
A. Titration of Transposase Concentration (with Fixed Time)
- Prepare Nuclei Suspension: Isolate and count nuclei. Dilute to a concentration of 10,000 nuclei in 50 µL of chilled lysis buffer (e.g., 10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl2, 0.1% IGEPAL CA-630).
- Set Up Reactions: Label 5 PCR tubes. To each tube, add 50 µL of nuclei suspension (10,000 nuclei). Keep on ice.
- Prepare Tagmentation Mix: Create a master mix containing 1X Tagmentation Buffer (provided with enzyme), and nuclease-free water. Aliquot this master mix into separate tubes, then add varying volumes of the commercial Tn5 transposase to achieve the following final volumes of enzyme per 50 µL total reaction: 1.25 µL, 2.5 µL (standard), 5 µL, 7.5 µL, 10 µL.
- Combine and Incubate: Add the respective tagmentation mixes to each nuclei tube. Mix gently by pipetting. Immediately transfer to a pre-warmed thermocycler at 37°C for 30 minutes.
- Purify DNA: Immediately add a stop solution (e.g., SDS or EDTA), then purify tagmented DNA using a MinElute PCR Purification Kit or SPRI beads. Elute in 21 µL of elution buffer.
- Library Amplification: Amplify each purified sample using a unique dual-indexing PCR kit (e.g., Nextera indexes) for 9-12 cycles. Purify the final library.
- QC Analysis: Run 1 µL of each library on a High Sensitivity Bioanalyzer/DNA TapeStation. Compare fragment distributions and molar yields.
B. Titration of Transposition Time (with Optimal Concentration)
- Based on results from Part A, choose the transposase volume yielding the best fragment distribution (peak ~200-600bp).
- Set Up Reactions: Prepare 4 identical reactions with the chosen enzyme volume, as in Part A.
- Variable Incubation: Incubate reactions at 37°C for 10 min, 30 min (standard), 60 min, and 90 min.
- Process and Analyze: Stop, purify, amplify, and QC libraries as in Part A steps 5-7. Assess the trade-off between yield/complexity and mitochondrial read percentage (requires qPCR for mitochondrial DNA or preliminary sequencing).
Visualization of Optimization Workflow and Decision Logic
Diagram 1: Two-Phase Optimization Workflow for ATAC-seq
Diagram 2: Effect of Transposition Parameters on Library Quality
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Transposition Optimization
| Item | Function in Optimization | Example Product/Catalog Number | Notes |
|---|---|---|---|
| Tn5 Transposase | Core enzyme for fragmentation and tagging. Varying this is the primary optimization variable. | Illumina Tagment DNA TDE1 Enzyme (20034197), or custom loaded Tn5. | Activity can vary between batches/commercial sources; consistency is key. |
| Tagmentation Buffer | Provides optimal ionic and chemical environment for Tn5 activity. | Illumina Tagment DNA Buffer (15027866). | Often used as a 2X concentrate. Do not alter during initial optimization. |
| Nuclei Isolation Reagents | To obtain clean, intact nuclei from the specific cell type. | IGEPAL CA-630 (I8896), Sucrose, MgCl2, Tris-HCl buffers. | Optimization starts with quality nuclei. Protocol varies by cell type (e.g., Omni-ATAC lysis buffer). |
| DNA Clean-up Beads | For efficient purification of tagmented DNA pre-amplification. | SPRIselect Beads (B23318), or equivalent PEG/SPRI beads. | Bead-to-sample ratio is critical for small fragment recovery. |
| High-Sensitivity DNA Assay Kit | Quantitative and qualitative analysis of pre- and post-amplification libraries. | Agilent High Sensitivity DNA Kit (5067-4626), Qubit dsDNA HS Assay. | Essential for measuring yield and visualizing fragment size distribution. |
| Indexed PCR Primers | To amplify tagmented DNA with unique dual indexes for multiplexing. | Illumina Nextera DNA CD Indexes (20018708), or IDT for Illumina Tagmentation. | Allows pooling of optimization samples for parallel sequencing assessment. |
| Real-Time PCR Master Mix | Optional, for quantifying mitochondrial DNA enrichment during time titration. | SYBR Green qPCR Master Mix. | Use primers for mitochondrial (e.g., MT-ND1) and nuclear (e.g., KRIT1) loci. |
Addressing Batch Effects and Ensuring Reproducibility Across Experiments
Within the broader thesis on a comprehensive ATAC-seq protocol step-by-step explanation, this whitepaper addresses the critical challenge of batch effects and reproducibility. As ATAC-seq becomes a cornerstone in epigenomic profiling for drug discovery and fundamental research, technical variability introduced across sample preparations, sequencing runs, and reagent lots threatens the validity of integrative analyses. This guide provides technical strategies to identify, mitigate, and control these factors.
Understanding Batch Effects in ATAC-seq
Batch effects are systematic technical differences between groups of samples processed separately. In ATAC-seq, they can arise from:
- Wet-lab Procedures: Variations in cell lysis, transposition efficiency, PCR amplification, and library preparation.
- Reagent Lots: Differences in enzyme activity (Tn5) or buffer composition between lots.
- Sequencing Runs: Differences in flow cell, chemistry, or cluster density.
- Personnel & Timing: Inter-operator variability and experiment drift over time.
These effects can confound biological signals, leading to false positives and irreproducible findings.
Quantitative Impact of Batch Effects
The following table summarizes common sources of batch effects and their measurable impact on ATAC-seq data quality.
Table 1: Common Sources of Batch Effects in ATAC-seq and Their Quantitative Impact
| Source Category | Specific Source | Typical Measurable Impact | Common Metric for Detection |
|---|---|---|---|
| Wet-Lab Protocol | Tn5 Transposition Time | +/- 15-30% in library complexity | FRiP score, Peak count, TSS enrichment |
| Cell Count Input | Major skew in insert size distribution; >50% variance in unique fragments | % of reads in peaks, Non-redundant fraction | |
| PCR Amplification Cycles | Duplication rate variance >20% | PCR bottleneck coefficient, Duplicate rate | |
| Reagent & Lot | Tn5 Enzyme Lot | Batch-correlated variance in global signal strength | Library complexity, Correlation (Pearson) between batches |
| DNA Purification Beads | Altered fragment size selection; efficiency variance +/- 10% | Fragment size distribution median | |
| Sequencing | Flow Cell/Lane | >5% difference in total read depth or cluster density | Total reads per sample, % Q30 bases |
| Sequencing Platform | Systematic differences in GC-bias profiles | GC content correlation across bins |
Methodologies for Batch Effect Detection and Correction
Experimental Design for Mitigation
- Randomization and Blocking: Process biological groups of interest across multiple batches. Do not confound batch with biological condition.
- Technical Replicates: Include control samples (e.g., reference cell line) in every batch to assess inter-batch variability.
- Balanced Library Pooling: Pool equimolar amounts of libraries from all experimental conditions for simultaneous sequencing.
Computational Detection Protocols
Protocol: Principal Component Analysis (PCA) for Batch Effect Screening
- Input: Normalized peak-by-sample count matrix (e.g., from DESeq2 or edgeR).
- Processing: Perform PCA on the matrix.
- Visualization: Plot the first 2-3 principal components, coloring points by both batch ID and biological condition.
- Interpretation: If samples cluster primarily by batch rather than condition in PC space, a significant batch effect is present.
Protocol: Using Negative Control Samples
- Include a set of identical reference samples (e.g., GM12878 cells) in every processing batch.
- After alignment and peak calling, calculate the correlation (e.g., Pearson) of signal in consensus peaks between all reference sample pairs.
- Plot a heatmap of the correlation matrix. High intra-batch and low inter-batch correlation indicates a batch effect.
Computational Correction Methods
- ComBat-seq (or ComBat): An empirical Bayes method implemented in the
svaR package designed for count-based NGS data. It adjusts for known batches while preserving biological signal. - Harmony: Integrates well with single-cell ATAC-seq data (scATAC-seq) to correct for embedding-level batch effects.
- Reference-Based Alignment: Align fragment counts from test samples to a stable reference dataset (e.g., from a large consortium) to remove systematic bias.
Essential Workflow for Reproducible ATAC-seq Analysis
ATAC-seq Reproducibility Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials and Reagents for Controlled ATAC-seq Experiments
| Item | Function & Importance for Reproducibility | Specification/Note |
|---|---|---|
| Validated Tn5 Transposase | Catalyzes simultaneous fragmentation and adapter tagging. Lot-to-lot variability is a major batch effect source. | Use commercially available, pre-loaded, QC'd kits (e.g., Illumina Tagment DNA TDE1). Aliquot and store at -80°C. |
| Cell Counting Standard | Accurate cell input (50K-100K) is critical for consistent chromatin complexity. | Use automated cell counters with calibrated protocols. Avoid hemaocytometers for high variability. |
| Reference Cell Line | Serves as an inter-batch control to monitor technical variation. | GM12878 (lymphoblastoid) is the ENCODE standard. Maintain consistent culture conditions. |
| DNA Purification Beads | For post-Tn5 cleanup and size selection. Bead-to-solution ratio affects size profile. | Use SPRISelect or equivalent. Calibrate the brand/ratio and do not switch mid-study. |
| High-Fidelity PCR Mix | Amplifies the library post-tagmentation. Enzyme fidelity affects duplicate rates. | Use a low-bias, proofreading polymerase mix (e.g., KAPA HiFi, NEB Next). |
| Dual-Indexed Adapters | Enables multiplexing of many samples for balanced pooling, reducing lane effects. | Use unique dual indexes (UDIs) to eliminate index hopping cross-talk. |
| Library Quantification Std. | Accurate molar quantification is essential for balanced pooling. | Use fluorometric assays (Qubit dsDNA HS) and fragment analyzer (Bioanalyzer/Tapestation). |
| Benchmarking Dataset | Public reference data for alignment and method comparison. | Use ENCODE ATAC-seq benchmarks (e.g., from Snyder lab) as a process control. |
Signaling Pathways Affected by Chromatin Accessibility
Chromatin Opening Drives Gene Expression
Addressing batch effects is not merely a computational afterthought but must be integrated into the experimental design, standardized protocol execution, and analytical pipeline of ATAC-seq research. By employing rigorous controls, balanced designs, and systematic detection/correction methods, researchers can ensure that observed differences in chromatin accessibility faithfully represent biology, thereby delivering reproducible and reliable insights for drug development and mechanistic studies.
This whitepaper, framed within a broader thesis on a step-by-step ATAC-seq protocol, addresses critical experimental challenges in chromatin accessibility profiling. While standard ATAC-seq requires >50,000 fresh cells, real-world research in translational medicine and drug development often involves limited, rare, or clinically preserved samples. This guide details advanced methodologies for robust ATAC-seq using low-input samples, frozen cells, and flash-frozen tissues, enabling studies on patient biopsies, sorted cell populations, and archival specimens.
Core Challenges and Optimization Principles
The primary hurdles when deviating from ideal fresh, high-cell-count samples include:
- Increased Nuclei Loss: From lysis and washing steps.
- Elevated Mitochondrial Contamination: Due to compromised nuclear envelopes in frozen samples.
- Background Noise: From suboptimal transposition or DNA damage.
- Sample Heterogeneity: Especially in tissue sections.
Optimizations focus on nuclei isolation, transposition efficiency, and library amplification.
Detailed Methodologies & Protocols
Protocol A: Low-Input ATAC-seq (500 - 5,000 Cells)
This protocol modifies the standard procedure to minimize loss.
- Cell Preparation: Harvest cells, centrifuge at 500 RCF for 5 min at 4°C. Resuspend gently in cold PBS.
- Lysis & Nuclei Preparation: Lyse cells in 50 µL of chilled Lysis Buffer (10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl2, 0.1% IGEPAL CA-630) for 3 minutes on ice. Critical: Immediately add 1 mL of Wash Buffer (10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl2) to stop lysis.
- Concentration: Pellet nuclei at 500 RCF for 10 min at 4°C. Carefully remove supernatant. Do not wash again. Proceed with transposition directly on the pellet.
- Scaled-Down Transposition: Resuspend nuclei pellet in 10 µL of Transposition Mix (from commercial kit, e.g., Illumina Tagmentase). Incubate at 37°C for 30 min in a thermomixer with agitation (1000 rpm).
- DNA Purification: Use a silica-membrane-based cleanup kit (e.g., MinElute PCR Purification Kit). Elute in 10 µL of Elution Buffer.
- Library Amplification: Amplify the entire eluate in a 25 µL PCR reaction using a high-fidelity, low-bias polymerase (e.g., KAPA HiFi HotStart ReadyMix). Determine cycle number via qPCR side-reaction or use a fixed 12-14 cycles for 500-5,000 cells.
- Double-Sided Size Selection: Clean up PCR reaction with 1.8x AMPure XP beads to remove large fragments. Transfer supernatant to new tube and add a further 0.5x beads to selectively bind fragments >150 bp. Elute in 20 µL.
Protocol B: ATAC-seq from Frozen Cell Pellet or Cryopreserved Tissue
This method prioritizes nuclei recovery from frozen material.
- Thawing: Rapidly thaw frozen cell pellet or ~1 mg tissue piece on ice in 1 mL of cold PBS + 0.1% BSA.
- Tissue Dissociation (if needed): For tissue, homogenize gently with a Dounce homogenizer (15-20 strokes with loose pestle) in Lysis Buffer.
- Nuclei Extraction & Fixation (Optional but Recommended): Resuspend pellet in 1 mL of Nuclear Extraction Buffer (NEB: 10 mM HEPES pH 7.9, 1.5 mM MgCl2, 10 mM KCl, 0.1% IGEPAL, 0.25 M Sucrose, 1x protease inhibitor). Incubate 5 min on ice. Layer suspension over 1 mL of Cushion Buffer (NEB with 0.35 M Sucrose, no IGEPAL). Centrifuge at 1300 RCF for 10 min at 4°C. This sucrose cushion improves purity.
- Nuclear Quality Assessment: Resuspend pellet in 50 µL PBS + 1% BSA. Stain with DAPI (1 µg/mL) and count/assess integrity via hemocytometer or flow cytometry. Expect lower yields than from fresh samples.
- Tagmentation & Library Prep: Follow Protocol A from Step 4, but consider increasing transposition time to 45-60 minutes to compensate for possible crosslinking from freezing.
Table 1: Performance Metrics Across Sample Types
| Sample Type | Recommended Cell Input | Typical Nuclei Yield Post-Lysis | Optimal Tagmentation Time (min) | Typical PCR Cycles | % Mitochondrial Reads (Post-Optimization) | Estimated Usable Sequencing Depth |
|---|---|---|---|---|---|---|
| Fresh, High-Quality Cells | 50,000+ | 45,000-48,000 | 30 | 8-10 | 10-30% | > 20M non-mt reads |
| Low-Input Fresh Cells | 500 - 5,000 | 400 - 4,500 | 30 | 12-14 | 20-50% | 5-15M non-mt reads |
| Frozen Cell Pellet | 20,000 - 50,000 | 10,000 - 30,000 | 45-60 | 12-15 | 30-70%* | 10-20M non-mt reads |
| Flash-Frozen Tissue | 1-10 mg | Highly Variable | 60 | 13-16 | 40-80%* | 5-15M non-mt reads |
*Can be significantly reduced via sucrose cushion centrifugation or post-sequencing computational filtering.
Table 2: Reagent Kits & Solutions for Optimized Workflows
| Reagent / Kit Name | Primary Function | Key Consideration for Challenging Samples |
|---|---|---|
| Tn5 Transposase (e.g., Illumina Tagmentase) | Simultaneous fragmentation and adapter tagging. | Use high-activity lots; for frozen samples, increase enzyme volume 1.2x and/or incubation time. |
| KAPA HiFi HotStart PCR Kit | High-fidelity library amplification with low GC bias. | Essential for limited DNA from low-input samples to prevent overcycling artifacts. |
| AMPure XP Beads | Solid-phase reversible immobilization (SPRI) for size selection. | Double-sided selection (e.g., 0.5x / 1.8x) is critical for removing primer dimer and large fragments. |
| Nuclear Extraction Buffer w/ Sucrose | Gentle, purified nuclei isolation. | The sucrose cushion step is critical for frozen samples to remove cytoplasmic debris and reduce mtDNA. |
| DAPI Stain | Fluorescent nuclei staining for quantification. | Vital for assessing nuclei integrity and accurately quantifying input pre-tagmentation. |
| Cell Lysis Buffer (IGEPAL-based) | Non-ionic detergent for plasma membrane lysis. | Concentration and time must be precisely controlled for small cell numbers to avoid over-lysis. |
Visualizations
Workflow for Frozen Sample ATAC-seq
Challenges & Targeted Optimizations Map
The Scientist's Toolkit: Essential Research Reagent Solutions
| Category | Item | Function & Rationale |
|---|---|---|
| Nuclei Isolation | IGEPAL CA-630 (10% stock) | Non-ionic detergent for controlled plasma membrane lysis; less harsh than NP-40, preserving nuclear integrity. |
| Sucrose Cushion Solution (0.35M Sucrose in NEB) | Density barrier to purify intact nuclei from cytoplasmic debris, critical for frozen samples to reduce mtDNA. | |
| Tagmentation | Custom-Loaded Tn5 Transposase | Enzyme pre-loaded with sequencing adapters. High-activity, batch-tested reagent is non-negotiable for low-input success. |
| Library Prep | MinElute PCR Purification Kit | Silica-membrane columns for efficient recovery of small DNA fragments (tagmented DNA) in low elution volumes (10 µL). |
| AMPure XP Beads | Magnetic beads for precise size selection. Double-sided cleanup removes both primer dimers (<100bp) and large contaminants. | |
| KAPA HiFi HotStart ReadyMix | Polymerase with high fidelity and low amplification bias, essential for even coverage from minimal template. | |
| QC & Assessment | DAPI (4',6-diamidino-2-phenylindole) | Fluorescent DNA stain for accurate counting and viability assessment of isolated nuclei via hemocytometer or flow cytometer. |
| Bioanalyzer/TapeStation HS DNA Chips | Microfluidic electrophoresis for precise library fragment size distribution analysis pre-sequencing. |
Validating ATAC-seq Data and Comparing it to Other Epigenomic Profiling Methods
This technical guide details the core bioinformatics pipeline for analyzing ATAC-seq (Assay for Transposase-Accessible Chromatin using sequencing) data, a pivotal component of a comprehensive thesis on the ATAC-seq protocol. The pipeline transforms raw sequencing reads into interpretable maps of chromatin accessibility, enabling researchers and drug development professionals to identify regulatory elements crucial for understanding gene expression dynamics in health and disease.
The Bioinformatics Pipeline: A Step-by-Step Technical Guide
Raw Data Assessment: FASTQ Quality Control
The initial step involves evaluating the quality of raw sequencing data stored in FASTQ files.
Experimental Protocol (FASTQ QC):
- Tool: FastQC is executed via the command line.
- Input: One or more
*.fastqor*.fastq.gzfiles. - Command:
fastqc sample_R1.fastq.gz sample_R2.fastq.gz -o ./qc_report/ - Output: An HTML report containing per-base sequence quality, adapter contamination, and overrepresented sequences.
- Post-QC: Use Trimmomatic or Cutadapt to trim low-quality bases and adapter sequences based on FastQC findings.
Read Alignment: Mapping to a Reference Genome
Trimmed reads are aligned to a reference genome to determine their genomic origin. The choice of aligner is critical for ATAC-seq due to its sensitivity to nucleosome positioning.
Detailed Aligner Comparison:
| Aligner | Key Algorithm | Best For ATAC-seq Because... | Typical Command (Paired-end) |
|---|---|---|---|
| Bowtie2 | FM-index, BWT | Speed, sensitivity, and well-established for short reads. | bowtie2 -x hg38 -1 sample_R1.fastq -2 sample_R2.fastq -S sample.sam |
| BWA-MEM | BWT & FM-index | Accuracy with longer reads and better gap handling. | bwa mem -t 8 hg38.fa sample_R1.fastq sample_R2.fastq > sample.sam |
| STAR | Spliced Alignment | Not recommended for standard ATAC-seq. Primarily for RNA-seq. | N/A |
Experimental Protocol (Alignment with Bowtie2):
- Index the Genome:
bowtie2-build hg38.fa hg38 - Perform Alignment: Use parameters to filter out poorly aligning reads. For ATAC-seq, it is common to allow soft-clipping but set a stringent MAPQ threshold.
- Post-Alignment Processing: Sort and index the BAM file using
samtools sortandsamtools index.
Post-Alignment Processing & Filtering
This step removes technical artifacts and identifies true open chromatin signals.
Key Filtering Steps:
- Remove Unmapped/Non-unique Reads: Use
samtools view -b -h -f 2 -F 1804 -q 30to keep properly paired, uniquely mapped reads. - Remove Mitochondrial Reads: ATAC-seq exhibits high mitochondrial DNA contamination. Remove chrM reads:
samtools idxstats sample.bam | cut -f 1 | grep -v chrM | xargs samtools view -b sample.bam > sample_noMito.bam - Mark Duplicates: Use Picard or
sambamba markdupto flag PCR duplicates, which can bias peak calling.
Quantitative Data on Filtering:
| Filtering Step | Typical % of Reads Removed (ATAC-seq) | Purpose |
|---|---|---|
| Low MAPQ/Non-unique | 10-30% | Eliminates ambiguous mappings |
| Mitochondrial Reads | 5-50% (Variable) | Removes uninformative, high-signal noise |
| PCR Duplicates | 5-30% | Prevents amplification bias |
Peak Calling: Identifying Open Chromatin Regions
Peak callers identify statistically significant regions of enriched read coverage (peaks), representing open chromatin.
Peak Caller Comparison:
| Peak Caller | Statistical Model | ATAC-seq Suitability | Key Consideration |
|---|---|---|---|
| MACS2 | Poisson/negative binomial | Excellent, widely adopted. | Use --nomodel --shift -100 --extsize 200 for ATAC-seq nucleosome-free fragments. |
| Genrich | Poisson | Excellent, designed for ATAC-seq. | Includes duplicate removal and auto-control from background. |
| HMMRATAC | Hidden Markov Model | Excellent, model-based. | Directly outputs nucleosome positions and footprints alongside peaks. |
Experimental Protocol (Peak Calling with MACS2):
- Input: Processed, filtered, and deduplicated BAM file.
- Command for ATAC-seq:
- Output:
*_peaks.narrowPeak(BED format),*_summits.bed, and signal tracks for visualization.
Workflow Visualization: ATAC-seq Bioinformatics Pipeline
Diagram Title: ATAC-seq Data Analysis Pipeline Workflow
The Scientist's Toolkit: Research Reagent Solutions
| Item / Solution | Function in ATAC-seq Protocol |
|---|---|
| Tn5 Transposase | Engineered enzyme that simultaneously fragments and tags accessible DNA with sequencing adapters. Core of the assay. |
| Nextera DNA Library Prep Kit | Commercial kit containing the Tn5 transposase and buffers for constructing sequencing-ready libraries. |
| AMPure XP Beads | Magnetic beads for size selection and purification of DNA fragments, crucial for selecting optimal fragment lengths. |
| Qubit dsDNA HS Assay Kit | Fluorometric quantification of library DNA concentration, more accurate for sequencing prep than UV spectrometry. |
| Bioanalyzer High Sensitivity DNA Kit | Microfluidics-based analysis to assess library fragment size distribution before sequencing. |
| PCR Reagents (NEB Next) | Enzymes and buffers for limited-cycle PCR to amplify the transposed DNA fragments and add full adapter sequences. |
| Dual Indexed Adapters (i7/i5) | Unique molecular barcodes for multiplexing multiple samples in a single sequencing run. |
| PhiX Control v3 | Spiked-in control library for monitoring sequencing run quality and balancing nucleotide diversity on Illumina flow cells. |
This guide is a component of a comprehensive thesis detailing the ATAC-seq (Assay for Transposase-Accessible Chromatin using sequencing) protocol. Following library preparation and sequencing, rigorous quality control (QC) is paramount. This document provides an in-depth technical analysis of three pivotal post-sequencing QC metrics used to assess the quality of ATAC-seq data: the Fraction of Reads in Peaks (FRiP) score, Transcription Start Site (TSS) Enrichment, and Fragment Size Distribution. Accurate assessment using these metrics is critical for researchers, scientists, and drug development professionals to ensure data integrity before proceeding with downstream biological interpretation.
Core Quality Control Metrics Explained
FRiP Score (Fraction of Reads in Peaks)
The FRiP score quantifies the signal-to-noise ratio in an ATAC-seq experiment. It is calculated as the proportion of all sequenced fragments (reads) that fall within identified peaks of open chromatin.
- Interpretation: A higher FRiP score indicates a more successful experiment with a strong, specific signal. Low FRiP scores suggest high background noise, potentially due to issues with cell viability, transposase activity, or over-digestion.
- Calculation:
FRiP = (Number of reads overlapping peak regions) / (Total number of mapped reads) - Benchmarks: As shown in Table 1, expected FRiP scores vary based on the biological sample and sequencing depth.
TSS Enrichment Score
This metric evaluates the enrichment of sequencing fragments around Transcription Start Sites (TSSs). Open chromatin is highly enriched at active promoters; thus, a successful ATAC-seq library should show a strong, phased signal centered on TSSs.
- Interpretation: The score is the ratio of the maximum read coverage near the TSS (±100 bp) to the average read coverage in a distal flanking region (e.g., ±2000-5000 bp). A high, sharp peak at the TSS is characteristic of high-quality data. A flattened profile suggests poor signal or excessive background.
- Benchmarks: A TSS enrichment score > 10 is generally considered good for human/mouse samples, though this can vary.
Fragment Size Distribution
The plot of fragment lengths (insert sizes) provides a fingerprint for ATAC-seq data. It reveals the periodicity of nucleosome positioning, as the transposase preferentially inserts into nucleosome-free regions.
- Interpretation:
- Peak < 100 bp: Represents nucleosome-free fragments (promoters, enhancers).
- Peak ~ 200 bp: Fragments protected by a single nucleosome.
- Peak ~ 400 bp: Fragments protected by two nucleosomes (di-nucleosome).
- Diagnostic Value: A clear, oscillatory pattern confirms proper enzymatic cleavage and library preparation. A dominant mononucleosome peak with minimal sub-nucleosomal (<100 bp) signal may indicate over-digestion.
Table 1: ATAC-seq QC Metric Benchmarks
| Metric | Definition | Recommended Range (Human/Mouse) | Threshold for Concern | Primary Indication |
|---|---|---|---|---|
| FRiP Score | Fraction of reads in called peaks. | 0.2 - 0.6 (cell lines); 0.1 - 0.3 (tissues) | < 0.1 | Low signal-to-noise ratio. |
| TSS Enrichment | Max coverage at TSS / avg. flank coverage. | > 10 | < 5 | Poor enrichment at active promoters. |
| Periodicity | Visibility of nucleosomal ladder in fragment plot. | Clear peaks at <100, ~200, ~400 bp. | No periodicity; single broad peak. | Failed reaction or over-fixation. |
Experimental Protocols for Metric Calculation
Protocol: Generating FRiP and TSS Enrichment Scores
This workflow assumes the starting point is paired-end FASTQ files.
Data Processing & Alignment:
- Use
fastporTrimmomaticto remove adapters and low-quality bases. - Align reads to the reference genome (e.g., hg38, mm10) using a splice-aware aligner like
Bowtie2orBWAin paired-end mode. - Use
samtoolsto sort and index the resulting BAM file. Mark duplicates usingpicard MarkDuplicates. - Filter the BAM file to retain only properly paired, non-duplicate, high-quality mapped reads.
- Use
Peak Calling (for FRiP):
- Call peaks using
MACS2with the--nomodeland--shift -100 --extsize 200parameters, which are tailored for ATAC-seq data. - Example command:
macs2 callpeak -t processed.bam -f BAMPE -n output_prefix --nomodel --shift -100 --extsize 200
- Call peaks using
FRiP Score Calculation:
- Using the filtered BAM file and the generated narrowPeak file, calculate the fraction of reads in peaks.
- Tools like
featureCounts(from Subread package) orbedtools intersectcan be used. bedtoolsexample:bedtools intersect -a filtered.bam -b peaks.narrowPeak -c | awk '{total++; inPeak+=$NF} END{print inPeak/total}'
TSS Enrichment Calculation:
- Generate a TSS enrichment profile and score using specialized tools.
- Using
deeptools: First, create a normalized bigWig file:bamCoverage -b filtered.bam -o coverage.bw --binSize 1 --normalizeUsing RPKM. - Compute the matrix around TSSs:
computeMatrix reference-point --referencePoint TSS -S coverage.bw -R genes.gtf -a 2000 -b 2000 -o matrix.gz. - The
plotProfileoutput will contain the TSS enrichment score.
Protocol: Visualizing Fragment Size Distribution
- Extract Insert Sizes:
- From the filtered, deduplicated BAM file, extract the fragment length (TLEN field) for all properly paired reads.
- Using
samtools:samtools view -F 0x04 -f 0x02 filtered.bam | awk '{print sqrt($9^2)}' > fragment_lengths.txt
- Plot Distribution:
- Import the list of fragment lengths into R or Python (Matplotlib) and generate a histogram.
- R/ggplot2 example code:
Visualizing the ATAC-seq QC Workflow
Diagram Title: ATAC-seq Data QC Analysis Pipeline
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Tools for ATAC-seq Library QC
| Item | Function / Role in QC | Example Product/Kit |
|---|---|---|
| High-Fidelity Transposase | Generates the library. Batch-to-batch consistency is critical for reproducible fragment size distributions. | Illumina Tagment DNA TDE1 Enzyme |
| AMPure XP Beads | Used for size selection and cleanup. Crucial for removing short fragments and adapter dimers that distort QC metrics. | Beckman Coulter AMPure XP |
| High-Sensitivity DNA Assay Kit | Quantifies library yield before sequencing. Ensures sufficient input for sequencing to achieve required depth for FRiP calculation. | Agilent Bioanalyzer HS DNA Kit / Qubit dsDNA HS Assay |
| Library Quantification Kit | Accurate quantification via qPCR for pooling and cluster generation on sequencer. | KAPA Library Quantification Kit |
| Sequence Alignment Software | Maps reads to genome for downstream analysis (BAM file generation). | Bowtie2, BWA-MEM, STAR |
| Peak Caller (ATAC-seq optimized) | Identifies open chromatin regions for FRiP score calculation. | MACS2 (with ATAC-seq mode) |
| QC & Visualization Tools | Calculates TSS enrichment, plots fragment size distributions, and generates metrics. | deepTools, SAMtools, bedtools, R/Bioconductor (ChIPQC) |
Within the broader thesis of ATAC-seq protocol optimization, robust validation of identified chromatin accessibility peaks is paramount. This guide details integrative bioinformatic and experimental strategies to validate ATAC-seq peaks by correlating them with orthogonal assays: DNase-seq (open chromatin), MNase-seq (nucleosome positioning), and key histone modification ChIP-seq marks. This multi-faceted approach establishes confidence in the biological relevance of accessibility data for downstream research and drug target discovery.
Foundational Principles of Cross-Assay Correlation
ATAC-seq uses hyperactive Tn5 transposase to tag accessible DNA. Validation involves demonstrating that these regions correspond to biologically meaningful chromatin states defined by other epigenomic profiles.
- DNase-seq Correlation: Expect high overlap, as both techniques identify open chromatin regions. Discrepancies may arise from technical differences (enzyme size, sequence bias) or biological context.
- MNase-seq Correlation: ATAC-seq peaks in open chromatin should align with MNase-sensitive (nucleosome-depleted) regions. Periodicity of insert sizes from ATAC-seq can further validate nucleosome positioning patterns.
- Histone Mark Correlation: Active regulatory elements (e.g., promoters, enhancers) identified by ATAC-seq should co-localize with permissive marks (H3K27ac, H3K4me3) and be devoid of repressive marks (H3K27me3).
Quantitative Data from Comparative Studies
Recent cross-assay studies provide benchmark expectations for peak overlap and correlation.
Table 1: Typical Peak Overlap Between ATAC-seq and Orthogonal Assays
| Assay Comparison | Typical Overlap Range | Key Influencing Factors |
|---|---|---|
| ATAC-seq vs. DNase-seq | 60-85% (for strong peaks) | Cell type, sequencing depth, peak-caller stringency, DNase I hypersensitivity site (DHS) definition. |
| ATAC-seq Open Regions vs. MNase-sensitive Zones | 70-90% | MNase digestion optimization, mononucleosome vs. subnucleosomal fragment analysis. |
| ATAC-seq Peaks at Active Promoters vs. H3K4me3 | >80% | Promoter definition window, mark specificity (H3K4me3 for promoters, H3K27ac for enhancers). |
| ATAC-seq Peaks at Enhancers vs. H3K27ac | 65-80% | Enhancer prediction method, cell-type specificity of the mark. |
Table 2: Recommended Bioinformatics Tools for Correlation Analysis
| Tool Name | Primary Function | Key Metric Output |
|---|---|---|
| BEDTools | Intersect genomic intervals (peaks). | Jaccard index, overlap counts. |
| deepTools | Generate correlation plots and heatmaps. | Pearson/Spearman correlation coefficients. |
| ChIPseeker | Annotate peaks and compare with genomic features. | Genomic distribution percentages. |
| IDR (Irreproducible Discovery Rate) | Assess reproducibility between replicates and across assays. | IDR score, ranked peak consistency. |
Experimental Protocols for Integrated Validation
Protocol 3.1: Computational Pipeline for Peak Overlap Analysis
- Data Acquisition: Download processed peak files (BED/narrowPeak format) for ATAC-seq and the corresponding validation assay (DNase-seq, MNase-seq, or ChIP-seq) from public repositories (ENCODE, GEO) or generate in-house.
- Peak Standardization: Convert all peak files to a unified genomic coordinate system (e.g., hg38). Use
BEDTools slopto standardize peak widths if necessary. Calculate Overlap: Use
BEDTools intersectto find overlapping genomic intervals. Apply a minimum reciprocal overlap threshold (e.g., 50%).Statistical Assessment: Compute the Jaccard index (size of intersection / size of union) and overlap percentages. Use IDR analysis for high-confidence peak sets.
Protocol 3.2: Genome-Wide Signal Correlation Workflow
- Generate Signal Files: Create genome-wide coverage bigWig files for each assay, normalized for sequencing depth (e.g., using Reads Per Kilobase per Million mapped reads - RPKM or CPM).
Compute Correlation Matrix: Using
deepTools multiBigwigSummary, calculate pairwise correlation values across the genome or at peak regions.Visualize: Plot the correlation matrix with
plotCorrelationor generate aggregate profile plots of signal intensity around ATAC-seq peak centers withplotProfile.
Visualization of Validation Strategies
ATAC-seq Peak Validation Strategy
Epigenetic Features at a Validated Accessible Region
Table 3: Key Reagents and Tools for Multi-Assay Validation Studies
| Item | Function in Validation Context | Example/Note |
|---|---|---|
| Hyperactive Tn5 Transposase | Core enzyme for ATAC-seq library prep. | Commercial kits (Illumina, Diagenode) ensure consistent activity. |
| DNase I (RNase-free) | For DNase-seq; cleaves accessible DNA. | Quality critical; requires precise titration. |
| Micrococcal Nuclease (MNase) | For MNase-seq; digests linker DNA between nucleosomes. | Requires optimization of digestion time/conc. for cell type. |
| Histone Modification Specific Antibodies | For ChIP-seq validation of active/repressive states. | Select antibodies with high ChIP-grade specificity (e.g., from CUT&Tag validated sets). |
| Next-Generation Sequencing Library Prep Kits | For constructing sequencing libraries from all assays. | Use compatible kits for low-input and high-throughput applications. |
| Size Selection Beads | Critical for isolating mononucleosomal (ATAC-seq, MNase-seq) or subnucleosomal fragments. | SPRI/AMPure beads allow precise size selection. |
| Cell Fixation Reagents (e.g., Formaldehyde) | For cross-linking in ChIP-seq protocols. | Cross-linking time must be optimized to balance signal and background. |
| Chromatin Shearing Device | For fragmenting cross-linked chromatin (ChIP-seq) or nuclei (MNase-seq). | Covaris sonicator or focused ultrasonicator for consistent fragment size. |
| High-Fidelity DNA Polymerase | For PCR amplification of sequencing libraries from all techniques. | Minimizes amplification bias and errors. |
| Bioinformatics Software Suites | For alignment, peak calling, and comparative analysis. | Use established pipelines (ENCODE ATAC-seq, DNase2ChIP) for consistency. |
1. Introduction
This analysis compares three pivotal methods for assaying chromatin accessibility and nucleosome positioning: Assay for Transposase-Accessible Chromatin with high-throughput sequencing (ATAC-seq), DNase I hypersensitive sites sequencing (DNase-seq), and Micrococcal Nuclease sequencing (MNase-seq). Framed within the context of advancing the ATAC-seq protocol, this guide details the operational principles, strengths, and limitations of each technique, providing researchers and drug development professionals with the information necessary to select the optimal method for their epigenetic investigations.
2. Core Methodologies and Experimental Protocols
2.1. ATAC-seq Protocol (Step-by-Step) ATAC-seq utilizes a hyperactive Tn5 transposase to simultaneously fragment and tag accessible genomic DNA with sequencing adapters.
- Cell Lysis & Transposition: Isolated nuclei are incubated with the Tn5 transposase loaded with sequencing adapters ("tagmentation"). The transposase inserts adapters into accessible DNA regions.
- DNA Purification: The tagmented DNA is purified using a silica-membrane column or SPRI beads.
- PCR Amplification: The purified DNA is amplified with barcoded primers for multiplexing. Library amplification is monitored using qPCR to avoid over-amplification.
- Library Purification & QC: The final library is purified and quantified via methods like qPCR, Bioanalyzer, or fragment analyzer before sequencing.
2.2. DNase-seq Protocol DNase-seq employs the DNase I enzyme to cleave exposed, nucleosome-depleted DNA.
- Nuclei Isolation & Digestion: Isolated nuclei are treated with a titrated amount of DNase I enzyme. The concentration is critical to achieve sparse, single cuts per hypersensitive site.
- Reaction Termination & DNA Extraction: Digestion is stopped with EDTA/SDS, and genomic DNA is extracted via phenol-chloroform.
- Size Selection & Adapter Ligation: Small DNA fragments (100-500 bp) are gel-purified, representing cleaved accessible sites. Blunt-ended fragments are ligated to sequencing adapters.
- Library Amplification & Sequencing: Ligation products are PCR-amplified and sequenced.
2.3. MNase-seq Protocol MNase-seq uses Micrococcal Nuclease to digest linker DNA between nucleosomes, mapping protected DNA.
- Cross-linking & Digestion: Chromatin is cross-linked with formaldehyde. Nuclei are isolated and digested with MNase, which preferentially cuts linker DNA.
- Reaction Termination & Decrosslinking: Digestion is stopped with EGTA/SDS, and crosslinks are reversed.
- Mononucleosome Isolation: DNA is purified and mononucleosomal fragments (~147 bp) are gel-extracted.
- Library Construction: Isolated DNA fragments are processed for standard Illumina library prep (end-repair, A-tailing, adapter ligation, PCR).
3. Comparative Analysis: Quantitative Data
Table 1: Technical Comparison of Chromatin Profiling Methods
| Feature | ATAC-seq | DNase-seq | MNase-seq |
|---|---|---|---|
| Core Principle | Transposase insertion into open chromatin | Endonuclease cleavage of open chromatin | Nuclease digestion of linker DNA |
| Starting Material | 50K - 100K cells (standard); <1K (optimized) | 0.5 - 50 million cells | 1 - 10 million cells |
| Primary Output | Open chromatin regions; nucleosome positions (periodicity) | DNase I Hypersensitive Sites (DHSs) | Nucleosome positions & occupancy; protected DNA |
| Resolution | Single-nucleotide (cut sites) | ~10-50 bp (cleavage clusters) | ~1-10 bp (nucleosome dyad) |
| Typical Sequencing Depth | 25 - 100 million reads | 100 - 300 million reads | 20 - 50 million reads |
| Assay Time | ~1 day (from cells to library) | 3-5 days | 2-4 days |
| Key Advantage | Fast, low input, maps TF footprints | Gold standard for in vivo hypersensitivity, robust footprinting | Gold standard for nucleosome positioning |
| Key Limitation | Mitochondrial read contamination, sensitive to transposon kinetics | High cell input, complex protocol, sequence bias of DNase I | Requires crosslinking (artifacts), indirect measure of accessibility |
Table 2: Functional Outputs and Detection Capabilities
| Capability | ATAC-seq | DNase-seq | MNase-seq |
|---|---|---|---|
| Maps Open Chromatin | Yes | Yes (High Sensitivity) | No (maps protected DNA) |
| Nucleosome Positioning | Yes (indirect, from fragment size) | No (clears nucleosomes) | Yes (Direct & High-Res) |
| TF Footprinting | Yes (moderate resolution) | Yes (High Resolution) | No |
| Sequence Bias | Low (minimal Tn5 sequence preference) | High (DNase I sequence preference) | Low (preference for AT-rich linkers) |
| Compatibility with Frozen Tissue | Yes (on nuclei) | Limited (requires fresh nuclei) | Yes (on crosslinked material) |
4. Visualized Workflows and Logical Relationships
Title: Comparative Workflows of Three Chromatin Assays
Title: Decision Guide for Selecting a Chromatin Assay
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials and Reagents
| Item | Function | Key Consideration |
|---|---|---|
| Hyperactive Tn5 Transposase (ATAC-seq) | Engineered enzyme for simultaneous fragmentation and adapter tagging of open chromatin. Core of ATAC-seq. | Commercial loaded kits (e.g., Illumina Tagment DNA TDE1) ensure reproducibility. Activity lot-to-lot variation can impact results. |
| DNase I, RNase-free (DNase-seq) | Endonuclease that cleaves DNA at accessible, protein-free regions. | Requires careful titration for optimal sparse cutting. Vendor and lot can affect sequence bias. |
| Micrococcal Nuclease (MNase-seq) | Endo-exonuclease that digests naked DNA, preferentially cutting linker DNA between nucleosomes. | Must be titrated to achieve >80% mononucleosomes. Calcium concentration is critical for activity. |
| SPRI Beads | Magnetic beads for size-selective purification and cleanup of DNA fragments in all protocols. | Ratio of beads to sample determines size cut-off. Essential for removing enzymes, salts, and small fragments. |
| PCR Barcoding Primers | Add unique sample indices and full sequencing adapters during library amplification for multiplexing. | Dual-indexed primers are standard to reduce index hopping artifacts in pooled sequencing. |
| Nuclear Prep Buffer (ATAC/DNase) | Lysis buffer designed to isolate intact nuclei while removing cytoplasmic content and nucleases. | Critical for ATAC-seq on cells. Must be optimized for tissue or frozen samples. Contains detergents like NP-40 or Digitonin. |
| Cell Permeabilization Reagent (e.g., Digitonin) | Used in ATAC-seq to permeabilize cells for transposase entry, enabling assay on intact cells. | Concentration is critical; too high damages nuclei, too low reduces efficiency. |
| Size Selection Method (e.g., Gel, Beads) | Isolates DNA fragments of specific size ranges (e.g., mononucleosomes for MNase-seq, cleaved fragments for DNase-seq). | Gel electrophoresis offers precise size selection; bead-based methods are faster and higher-throughput. |
6. Conclusion
ATAC-seq, DNase-seq, and MNase-seq offer complementary views of chromatin architecture. ATAC-seq excels in speed, low input requirements, and simultaneous mapping of accessibility and nucleosome positioning, driving its rapid adoption in single-cell and large-scale population studies. DNase-seq remains the gold standard for high-resolution mapping of in vivo DNase hypersensitive sites and transcription factor footprints, albeit with greater sample demands. MNase-seq is the definitive method for interrogating nucleosome positioning and occupancy. The choice of assay must be guided by the specific biological question, available sample type and quantity, and required resolution, with ongoing protocol refinements continuing to expand the applications and robustness of each technique.
Integrating ATAC-seq with RNA-seq and ChIP-seq for Multi-Omics Insights
This technical guide details the methodology for integrating ATAC-seq with RNA-seq and ChIP-seq data, framed within broader research on the ATAC-seq protocol. The convergence of these assays provides a systems-level view of chromatin accessibility, gene expression, and transcription factor binding or histone modifications, enabling the deconvolution of gene regulatory networks critical for understanding disease mechanisms and identifying therapeutic targets.
Foundational Assay Principles & Quantitative Outputs
Each omics layer generates distinct but complementary quantitative data. Key metrics are summarized below.
Table 1: Core Outputs and Metrics from Individual Assays
| Assay | Primary Output | Key Quantitative Metrics | Typical Resolution/Scale |
|---|---|---|---|
| ATAC-seq | Genome-wide chromatin accessibility landscape | Peak count, insert size distribution, TSS enrichment score, fragment length periodicity, read depth in peaks. | Nucleosome resolution (~200 bp peaks). |
| RNA-seq | Genome-wide transcript abundance | Reads per gene (FPKM, TPM), differential expression (log2FC, p-value), splicing events (PSI). | Single gene/transcript. |
| ChIP-seq | Protein-DNA interaction sites (TFs or histones) | Peak count, peak score (-log10(p-value)), fold enrichment over control, motif occurrence. | 100-1000 bp regions. |
Integrated Multi-Omics Analysis Workflow
A successful integration requires coordinated experimental design and a structured bioinformatics pipeline.
Experimental Design Protocol:
- Cell/Tissue Source: Use biologically matched samples for all assays. Replicates (n≥3) are non-negotiable for statistical rigor.
- Sequencing Depth: Follow current field standards:
- ATAC-seq: 50-100 million paired-end reads per sample (e.g., 2x50 bp or 2x75 bp).
- RNA-seq: 30-50 million reads per sample for expression; more for isoform analysis.
- ChIP-seq: 20-40 million reads per sample (dependent on antigen).
- Data Generation Order: Process samples for all omics layers in parallel to minimize batch effects. If sequential, ensure proper sample cryopreservation.
Core Computational Integration Methodology:
- Step 1: Individual Assay Processing.
- ATAC-seq: Align reads (e.g., BWA-MEM2, Bowtie2). Remove mitochondrial reads and PCR duplicates. Call peaks (e.g., MACS2, Genrich). Generate bigWig files for visualization (normalized by sequencing depth).
- RNA-seq: Align to transcriptome (STAR, HISAT2). Quantify gene/isoform counts (featureCounts, Salmon). Perform differential expression analysis (DESeq2, edgeR).
- ChIP-seq: Align reads, remove duplicates. Call peaks relative to input control (MACS2, SICER). Identify enriched motifs (HOMER, MEME-ChIP).
- Step 2: Data Alignment and Correlation.
- Genomic coordinates from ATAC-seq and ChIP-seq are mapped to gene annotations (e.g., using
ChIPseekerin R/Bioconductor). - Correlate ATAC-seq signal intensity at promoter/enhancer regions with expression of putative target genes from RNA-seq. Statistical tests (e.g., linear regression) assess significance.
- Genomic coordinates from ATAC-seq and ChIP-seq are mapped to gene annotations (e.g., using
- Step 3: Integrative Insight Generation.
- Cis-regulatory Element (CRE) Linking: Overlap ATAC-seq peaks (accessible regions) with ChIP-seq peaks (TF binding sites). Use tools like BEDTools.
- Regulatory Network Inference: Link integrated CREs (ATAC+ChIP) to target genes (RNA-seq) based on proximity or chromatin conformation data (e.g., Hi-C). Use tools like
r3CseqorCytoscapefor network visualization. - Trajectory Analysis: For time-series or perturbation studies, use tools like
Monocle3orCiceroto model coordinated changes in accessibility and expression.
Diagram 1: Multi-omics data integration workflow.
Case Study: Identifying an Oncogenic Regulatory Circuit
Objective: Discover a transcription factor (TF) driving tumor progression via chromatin remodeling and target gene activation. Protocol:
- Data Generation: Perform ATAC-seq, RNA-seq, and TF ChIP-seq (e.g., for MYC) on matched tumor and normal cell lines.
- Differential Analysis:
- Identify Differentially Accessible Regions (DARs) from ATAC-seq (
DESeq2on peak counts). - Identify Differentially Expressed Genes (DEGs) from RNA-seq.
- Identify differential TF binding sites from ChIP-seq.
- Identify Differentially Accessible Regions (DARs) from ATAC-seq (
- Integration:
- Intersect DARs with differential MYC binding sites using
BEDTools intersect. This yields "gained" MYC sites in newly accessible chromatin. - Link these integrated sites to nearby (<100kb) up-regulated DEGs. Perform enrichment analysis (GREAT).
- Intersect DARs with differential MYC binding sites using
- Validation:
- Select top candidate target gene.
- CRISPRi/k.o. of MYC: Repeat ATAC-seq and RNA-seq to confirm loss of accessibility and expression at the candidate enhancer-gene pair.
- Reporter Assay: Clone the candidate accessible MYC-bound region into a luciferase vector. Co-transfect with MYC expression plasmid to test enhancer activity.
Diagram 2: Oncogenic TF circuit revealed by multi-omics.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Kits for Integrated Multi-Omics Studies
| Item | Function | Example Vendor/Cat. # (Illustrative) |
|---|---|---|
| Nextera DNA Library Prep Kit | Prepares sequencing-ready libraries from ATAC-seq tagmented DNA. | Illumina, FC-121-1030 |
| Tn5 Transposase (Tagmentase) | Engineered transposase simultaneously fragments and tags chromatin DNA with sequencing adapters. | Illumina, 20034197 |
| Dynabeads Protein A/G | Magnetic beads for antibody capture in ChIP-seq protocol. | Thermo Fisher, 10002D / 10004D |
| NEBNext Ultra II RNA Library Prep | High-efficiency library preparation for RNA-seq. | NEB, E7770 |
| SPRIselect Beads | Size selection and clean-up for all library types. | Beckman Coulter, B23318 |
| Validated ChIP-seq Grade Antibody | Antibody with high specificity and efficacy for ChIP. | Cell Signaling Tech., Abcam |
| RNase Inhibitor | Protects RNA integrity during RNA-seq library prep. | Takara, 2313A |
| DAPI or SYBR Green I | Cell cycle/dosing quantification for ATAC-seq cell counting. | Thermo Fisher, D1306 / S7585 |
| PCR Amplification Kit (High-Fidelity) | Amplifies limited material post-ChIP or ATAC tagmentation. | KAPA HiFi HotStart, KK2602 |
| Dual-index Barcode Adapters | Enables multiplexing of samples from different assays. | Illumina, 20022370 |
Advanced Integrative Analysis & Tools
Table 3: Software Packages for Multi-Omics Integration
| Tool Name | Primary Function | Language/Platform |
|---|---|---|
| ArchR | Integrative analysis of ATAC-seq and RNA-seq for single-cell and bulk data. | R |
| MEME Suite | Discovers enriched motifs in ATAC-seq/ChIP-seq peaks and links TFs to targets. | Command Line / Web |
| DiffBind | Differential binding analysis for ChIP-seq/ATAC-seq peak sets. | R/Bioconductor |
| IGV (Integrative Genomics Viewer) | Visualizes aligned read coverage and peaks from all three assays simultaneously. | Java |
| Cistrome | Toolkit for ChIP-seq & ATAC-seq analysis; includes pipeline for integration. | Pipeline/Galaxy |
| LIMMA | Fits linear models to integrate and test associations between different omics datasets. | R/Bioconductor |
Conclusion
Mastering the ATAC-seq protocol from bench to bioinformatics empowers researchers to robustly map the dynamic landscape of chromatin accessibility, a critical layer of epigenetic regulation. By understanding its foundational principles, executing a meticulous step-by-step protocol, proactively troubleshooting, and rigorously validating results against complementary methods, scientists can generate high-quality data to uncover novel regulatory elements, transcription factor binding sites, and nucleosome positions. As the field advances, the integration of ATAC-seq with single-cell technologies, spatial omics, and long-read sequencing promises even deeper insights into cellular heterogeneity and disease mechanisms. This positions ATAC-seq as an indispensable tool for driving discovery in fundamental biology, identifying therapeutic targets, and advancing personalized medicine.